9.5 Types of Body Movements
Learning objectives.
By the end of this section, you will be able to:
- Define the different types of body movements
- Identify the joints that allow for these motions
Synovial joints allow the body a tremendous range of movements. Each movement at a synovial joint results from the contraction or relaxation of the muscles that are attached to the bones on either side of the articulation. The type of movement that can be produced at a synovial joint is determined by its structural type. While the ball-and-socket joint gives the greatest range of movement at an individual joint, in other regions of the body, several joints may work together to produce a particular movement. Overall, each type of synovial joint is necessary to provide the body with its great flexibility and mobility. There are many types of movement that can occur at synovial joints ( Table 9.1 ). Movement types are generally paired, with one being the opposite of the other. Body movements are always described in relation to the anatomical position of the body: upright stance, with upper limbs to the side of body and palms facing forward. Refer to Figure 9.12 as you go through this section.

Interactive Link
Watch this video to learn about anatomical motions. What motions involve increasing or decreasing the angle of the foot at the ankle?
Flexion and Extension
Flexion and extension are typically movements that take place within the sagittal plane and involve anterior or posterior movements of the neck, trunk, or limbs. For the vertebral column, flexion (anterior flexion) is an anterior (forward) bending of the neck or trunk, while extension involves a posterior-directed motion, such as straightening from a flexed position or bending backward. Lateral flexion of the vertebral column occurs in the coronal plane and is defined as the bending of the neck or trunk toward the right or left side. These movements of the vertebral column involve both the symphysis joint formed by each intervertebral disc, as well as the plane type of synovial joint formed between the inferior articular processes of one vertebra and the superior articular processes of the next lower vertebra.
In the limbs, flexion decreases the angle between the bones (bending of the joint), while extension increases the angle and straightens the joint. For the upper limb, all anterior-going motions are flexion and all posterior-going motions are extension. These include anterior-posterior movements of the arm at the shoulder, the forearm at the elbow, the hand at the wrist, and the fingers at the metacarpophalangeal and interphalangeal joints. For the thumb, extension moves the thumb away from the palm of the hand, within the same plane as the palm, while flexion brings the thumb back against the index finger or into the palm. These motions take place at the first carpometacarpal joint. In the lower limb, bringing the thigh forward and upward is flexion at the hip joint, while any posterior-going motion of the thigh is extension. Note that extension of the thigh beyond the anatomical (standing) position is greatly limited by the ligaments that support the hip joint. Knee flexion is the bending of the knee to bring the foot toward the posterior thigh, and extension is the straightening of the knee. Flexion and extension movements are seen at the hinge, condyloid, saddle, and ball-and-socket joints of the limbs (see Figure 9.12 a-d ).
Hyperextension is the abnormal or excessive extension of a joint beyond its normal range of motion, thus resulting in injury. Similarly, hyperflexion is excessive flexion at a joint. Hyperextension injuries are common at hinge joints such as the knee or elbow. In cases of “whiplash” in which the head is suddenly moved backward and then forward, a patient may experience both hyperextension and hyperflexion of the cervical region.
Abduction and Adduction
Abduction and adduction motions occur within the coronal plane and involve medial-lateral motions of the limbs, fingers, toes, or thumb. Abduction moves the limb laterally away from the midline of the body, while adduction is the opposing movement that brings the limb toward the body or across the midline. For example, abduction is raising the arm at the shoulder joint, moving it laterally away from the body, while adduction brings the arm down to the side of the body. Similarly, abduction and adduction at the wrist moves the hand away from or toward the midline of the body. Spreading the fingers or toes apart is also abduction, while bringing the fingers or toes together is adduction. For the thumb, abduction is the anterior movement that brings the thumb to a 90° perpendicular position, pointing straight out from the palm. Adduction moves the thumb back to the anatomical position, next to the index finger. Abduction and adduction movements are seen at condyloid, saddle, and ball-and-socket joints (see Figure 9.12 e ).
Circumduction
Circumduction is the movement of a body region in a circular manner, in which one end of the body region being moved stays relatively stationary while the other end describes a circle. It involves the sequential combination of flexion, adduction, extension, and abduction at a joint. This type of motion is found at biaxial condyloid and saddle joints, and at multiaxial ball-and-sockets joints (see Figure 9.12 e ).
Rotation can occur within the vertebral column, at a pivot joint, or at a ball-and-socket joint. Rotation of the neck or body is the twisting movement produced by the summation of the small rotational movements available between adjacent vertebrae. At a pivot joint, one bone rotates in relation to another bone. This is a uniaxial joint, and thus rotation is the only motion allowed at a pivot joint. For example, at the atlantoaxial joint, the first cervical (C1) vertebra (atlas) rotates around the dens, the upward projection from the second cervical (C2) vertebra (axis). This allows the head to rotate from side to side as when shaking the head “no.” The proximal radioulnar joint is a pivot joint formed by the head of the radius and its articulation with the ulna. This joint allows for the radius to rotate along its length during pronation and supination movements of the forearm.
Rotation can also occur at the ball-and-socket joints of the shoulder and hip. Here, the humerus and femur rotate around their long axis, which moves the anterior surface of the arm or thigh either toward or away from the midline of the body. Movement that brings the anterior surface of the limb toward the midline of the body is called medial (internal) rotation . Conversely, rotation of the limb so that the anterior surface moves away from the midline is lateral (external) rotation (see Figure 9.12 f ). Be sure to distinguish medial and lateral rotation, which can only occur at the multiaxial shoulder and hip joints, from circumduction, which can occur at either biaxial or multiaxial joints.
Supination and Pronation
Supination and pronation are movements of the forearm. In the anatomical position, the upper limb is held next to the body with the palm facing forward. This is the supinated position of the forearm. In this position, the radius and ulna are parallel to each other. When the palm of the hand faces backward, the forearm is in the pronated position , and the radius and ulna form an X-shape.
Supination and pronation are the movements of the forearm that go between these two positions. Pronation is the motion that moves the forearm from the supinated (anatomical) position to the pronated (palm backward) position. This motion is produced by rotation of the radius at the proximal radioulnar joint, accompanied by movement of the radius at the distal radioulnar joint. The proximal radioulnar joint is a pivot joint that allows for rotation of the head of the radius. Because of the slight curvature of the shaft of the radius, this rotation causes the distal end of the radius to cross over the distal ulna at the distal radioulnar joint. This crossing over brings the radius and ulna into an X-shape position. Supination is the opposite motion, in which rotation of the radius returns the bones to their parallel positions and moves the palm to the anterior facing (supinated) position. It helps to remember that supination is the motion you use when scooping up soup with a spoon (see Figure 9.13 g ).
Dorsiflexion and Plantar Flexion
Dorsiflexion and plantar flexion are movements at the ankle joint, which is a hinge joint. Lifting the front of the foot, so that the top of the foot moves toward the anterior leg is dorsiflexion, while lifting the heel of the foot from the ground or pointing the toes downward is plantar flexion. These are the only movements available at the ankle joint (see Figure 9.13 h ).
Inversion and Eversion
Inversion and eversion are complex movements that involve the multiple plane joints among the tarsal bones of the posterior foot (intertarsal joints) and thus are not motions that take place at the ankle joint. Inversion is the turning of the foot to angle the bottom of the foot toward the midline, while eversion turns the bottom of the foot away from the midline. The foot has a greater range of inversion than eversion motion. These are important motions that help to stabilize the foot when walking or running on an uneven surface and aid in the quick side-to-side changes in direction used during active sports such as basketball, racquetball, or soccer (see Figure 9.13 i ).
Protraction and Retraction
Protraction and retraction are anterior-posterior movements of the scapula or mandible. Protraction of the scapula occurs when the shoulder is moved forward, as when pushing against something or throwing a ball. Retraction is the opposite motion, with the scapula being pulled posteriorly and medially, toward the vertebral column. For the mandible, protraction occurs when the lower jaw is pushed forward, to stick out the chin, while retraction pulls the lower jaw backward. (See Figure 9.13 j .)
Depression and Elevation
Depression and elevation are downward and upward movements of the scapula or mandible. The upward movement of the scapula and shoulder is elevation, while a downward movement is depression. These movements are used to shrug your shoulders. Similarly, elevation of the mandible is the upward movement of the lower jaw used to close the mouth or bite on something, and depression is the downward movement that produces opening of the mouth (see Figure 9.13 k ).
Excursion is the side to side movement of the mandible. Lateral excursion moves the mandible away from the midline, toward either the right or left side. Medial excursion returns the mandible to its resting position at the midline.
Superior Rotation and Inferior Rotation
Superior and inferior rotation are movements of the scapula and are defined by the direction of movement of the glenoid cavity. These motions involve rotation of the scapula around a point inferior to the scapular spine and are produced by combinations of muscles acting on the scapula. During superior rotation , the glenoid cavity moves upward as the medial end of the scapular spine moves downward. This is a very important motion that contributes to upper limb abduction. Without superior rotation of the scapula, the greater tubercle of the humerus would hit the acromion of the scapula, thus preventing any abduction of the arm above shoulder height. Superior rotation of the scapula is thus required for full abduction of the upper limb. Superior rotation is also used without arm abduction when carrying a heavy load with your hand or on your shoulder. You can feel this rotation when you pick up a load, such as a heavy book bag and carry it on only one shoulder. To increase its weight-bearing support for the bag, the shoulder lifts as the scapula superiorly rotates. Inferior rotation occurs during limb adduction and involves the downward motion of the glenoid cavity with upward movement of the medial end of the scapular spine.
Opposition and Reposition
Opposition is the thumb movement that brings the tip of the thumb in contact with the tip of a finger. This movement is produced at the first carpometacarpal joint, which is a saddle joint formed between the trapezium carpal bone and the first metacarpal bone. Thumb opposition is produced by a combination of flexion and abduction of the thumb at this joint. Returning the thumb to its anatomical position next to the index finger is called reposition (see Figure 9.13 l ).
This book may not be used in the training of large language models or otherwise be ingested into large language models or generative AI offerings without OpenStax's permission.
Want to cite, share, or modify this book? This book uses the Creative Commons Attribution License and you must attribute OpenStax.
Access for free at https://openstax.org/books/anatomy-and-physiology-2e/pages/1-introduction
- Authors: J. Gordon Betts, Kelly A. Young, James A. Wise, Eddie Johnson, Brandon Poe, Dean H. Kruse, Oksana Korol, Jody E. Johnson, Mark Womble, Peter DeSaix
- Publisher/website: OpenStax
- Book title: Anatomy and Physiology 2e
- Publication date: Apr 20, 2022
- Location: Houston, Texas
- Book URL: https://openstax.org/books/anatomy-and-physiology-2e/pages/1-introduction
- Section URL: https://openstax.org/books/anatomy-and-physiology-2e/pages/9-5-types-of-body-movements
© Jun 13, 2024 OpenStax. Textbook content produced by OpenStax is licensed under a Creative Commons Attribution License . The OpenStax name, OpenStax logo, OpenStax book covers, OpenStax CNX name, and OpenStax CNX logo are not subject to the Creative Commons license and may not be reproduced without the prior and express written consent of Rice University.
Module 10: Joints
Types of body movements, learning objectives.
- Define the different types of body movements
- Identify the joints that allow for these motions
Synovial joints allow the body a tremendous range of movements. Each movement at a synovial joint results from the contraction or relaxation of the muscles that are attached to the bones on either side of the articulation. The type of movement that can be produced at a synovial joint is determined by its structural type. While the ball-and-socket joint gives the greatest range of movement at an individual joint, in other regions of the body, several joints may work together to produce a particular movement. Overall, each type of synovial joint is necessary to provide the body with its great flexibility and mobility. There are many types of movement that can occur at synovial joints (Table 1). Movement types are generally paired, with one being the opposite of the other. Body movements are always described in relation to the anatomical position of the body: upright stance, with upper limbs to the side of body and palms facing forward.
Watch this video to learn about anatomical motions. What motions involve increasing or decreasing the angle of the foot at the ankle?
Flexion and Extension
Flexion and extension are movements that take place within the sagittal plane and involve anterior or posterior movements of the body or limbs. For the vertebral column, flexion (anterior flexion) is an anterior (forward) bending of the neck or body, while extension involves a posterior-directed motion, such as straightening from a flexed position or bending backward. Lateral flexion is the bending of the neck or body toward the right or left side. These movements of the vertebral column involve both the symphysis joint formed by each intervertebral disc, as well as the plane type of synovial joint formed between the inferior articular processes of one vertebra and the superior articular processes of the next lower vertebra.
In the limbs, flexion decreases the angle between the bones (bending of the joint), while extension increases the angle and straightens the joint. For the upper limb, all anterior-going motions are flexion and all posterior-going motions are extension. These include anterior-posterior movements of the arm at the shoulder, the forearm at the elbow, the hand at the wrist, and the fingers at the metacarpophalangeal and interphalangeal joints. For the thumb, extension moves the thumb away from the palm of the hand, within the same plane as the palm, while flexion brings the thumb back against the index finger or into the palm. These motions take place at the first carpometacarpal joint. In the lower limb, bringing the thigh forward and upward is flexion at the hip joint, while any posterior-going motion of the thigh is extension. Note that extension of the thigh beyond the anatomical (standing) position is greatly limited by the ligaments that support the hip joint. Knee flexion is the bending of the knee to bring the foot toward the posterior thigh, and extension is the straightening of the knee. Flexion and extension movements are seen at the hinge, condyloid, saddle, and ball-and-socket joints of the limbs (see Figure 1).
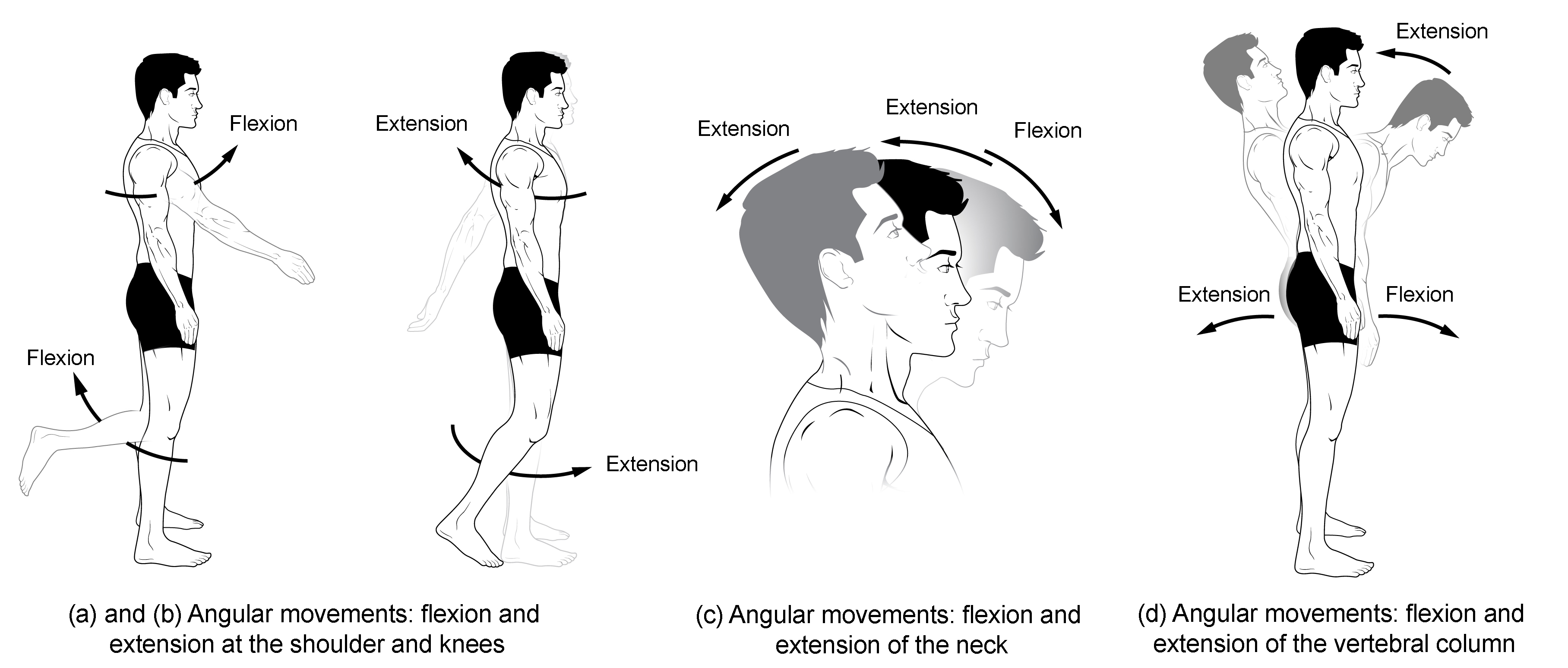
Figure 1. Flexion and extension. (a)–(b) Flexion and extension motions are in the sagittal (anterior–posterior) plane of motion. These movements take place at the shoulder, hip, elbow, knee, wrist, metacarpophalangeal, metatarsophalangeal, and interphalangeal joints. (c)–(d) Anterior bending of the head or vertebral column is flexion, while any posterior-going movement is extension.
Hyperextension is the abnormal or excessive extension of a joint beyond its normal range of motion, thus resulting in injury. Similarly, hyperflexion is excessive flexion at a joint. Hyperextension injuries are common at hinge joints such as the knee or elbow. In cases of “whiplash” in which the head is suddenly moved backward and then forward, a patient may experience both hyperextension and hyperflexion of the cervical region.
Abduction, Adduction, and Circumduction
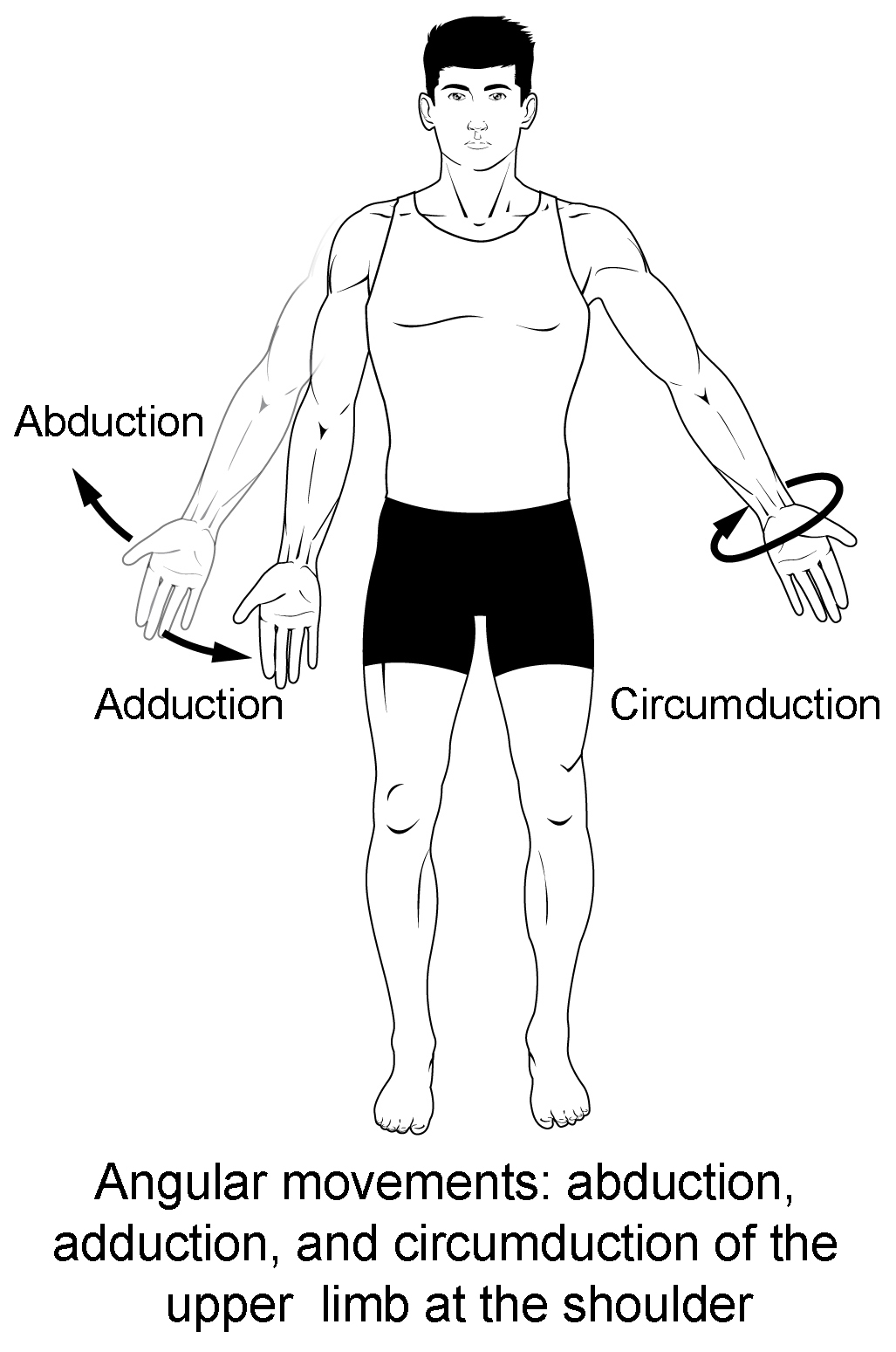
Figure 2. Abduction, adduction, and circumduction.
Abduction and adduction are motions of the limbs, hand, fingers, or toes in the coronal (medial–lateral) plane of movement. Moving the limb or hand laterally away from the body, or spreading the fingers or toes, is abduction. Adduction brings the limb or hand toward or across the midline of the body, or brings the fingers or toes together. Circumduction is the movement of the limb, hand, or fingers in a circular pattern, using the sequential combination of flexion, adduction, extension, and abduction motions.
Adduction, abduction, and circumduction take place at the shoulder, hip, wrist, metacarpophalangeal, and metatarsophalangeal joints.
Abduction and Adduction
Abduction and adduction motions occur within the coronal plane and involve medial-lateral motions of the limbs, fingers, toes, or thumb. Abduction moves the limb laterally away from the midline of the body, while adduction is the opposing movement that brings the limb toward the body or across the midline. For example, abduction is raising the arm at the shoulder joint, moving it laterally away from the body, while adduction brings the arm down to the side of the body. Similarly, abduction and adduction at the wrist moves the hand away from or toward the midline of the body. Spreading the fingers or toes apart is also abduction, while bringing the fingers or toes together is adduction. For the thumb, abduction is the anterior movement that brings the thumb to a 90° perpendicular position, pointing straight out from the palm. Adduction moves the thumb back to the anatomical position, next to the index finger. Abduction and adduction movements are seen at condyloid, saddle, and ball-and-socket joints (see Figure 2).
Circumduction
Circumduction is the movement of a body region in a circular manner, in which one end of the body region being moved stays relatively stationary while the other end describes a circle. It involves the sequential combination of flexion, adduction, extension, and abduction at a joint. This type of motion is found at biaxial condyloid and saddle joints, and at multiaxial ball-and-sockets joints (see Figure 2).
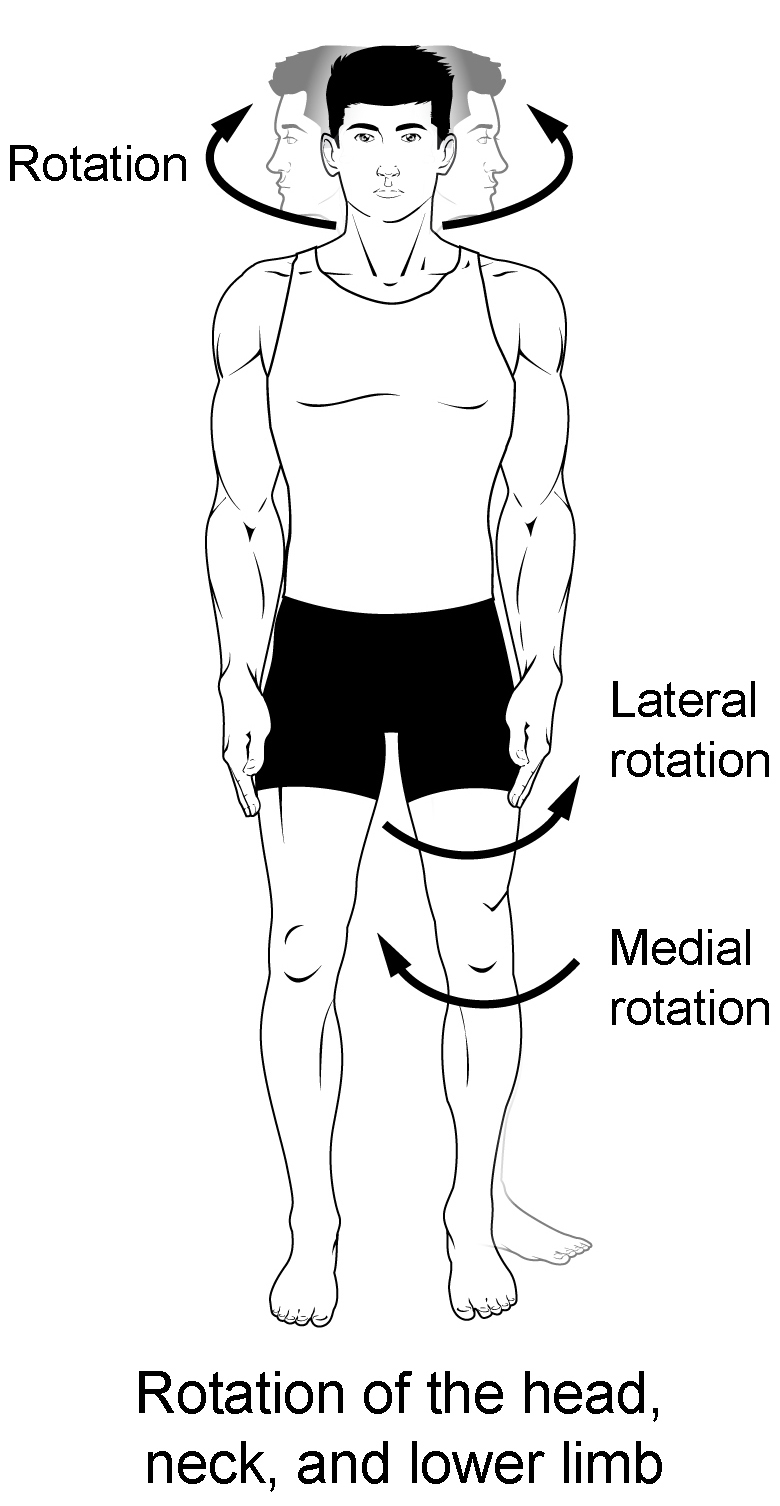
Figure 3. Rotation.
Rotation can occur within the vertebral column, at a pivot joint, or at a ball-and-socket joint. Rotation of the neck or body is the twisting movement produced by the summation of the small rotational movements available between adjacent vertebrae. At a pivot joint, one bone rotates in relation to another bone. This is a uniaxial joint, and thus rotation is the only motion allowed at a pivot joint. For example, at the atlantoaxial joint, the first cervical (C1) vertebra (atlas) rotates around the dens, the upward projection from the second cervical (C2) vertebra (axis). This allows the head to rotate from side to side as when shaking the head “no.” The proximal radioulnar joint is a pivot joint formed by the head of the radius and its articulation with the ulna. This joint allows for the radius to rotate along its length during pronation and supination movements of the forearm.
Rotation can also occur at the ball-and-socket joints of the shoulder and hip. Here, the humerus and femur rotate around their long axis, which moves the anterior surface of the arm or thigh either toward or away from the midline of the body. Movement that brings the anterior surface of the limb toward the midline of the body is called medial (internal) rotation . Conversely, rotation of the limb so that the anterior surface moves away from the midline is lateral (external) rotation (see Figure 3). Be sure to distinguish medial and lateral rotation, which can only occur at the multiaxial shoulder and hip joints, from circumduction, which can occur at either biaxial or multiaxial joints.
Turning of the head side to side or twisting of the body is rotation. Medial and lateral rotation of the upper limb at the shoulder or lower limb at the hip involves turning the anterior surface of the limb toward the midline of the body (medial or internal rotation) or away from the midline (lateral or external rotation).
Supination and Pronation
Supination and pronation are movements of the forearm. In the anatomical position, the upper limb is held next to the body with the palm facing forward. This is the supinated position of the forearm. In this position, the radius and ulna are parallel to each other. When the palm of the hand faces backward, the forearm is in the pronated position , and the radius and ulna form an X-shape.
Supination and pronation are the movements of the forearm that go between these two positions. Pronation is the motion that moves the forearm from the supinated (anatomical) position to the pronated (palm backward) position. This motion is produced by rotation of the radius at the proximal radioulnar joint, accompanied by movement of the radius at the distal radioulnar joint. The proximal radioulnar joint is a pivot joint that allows for rotation of the head of the radius. Because of the slight curvature of the shaft of the radius, this rotation causes the distal end of the radius to cross over the distal ulna at the distal radioulnar joint. This crossing over brings the radius and ulna into an X-shape position. Supination is the opposite motion, in which rotation of the radius returns the bones to their parallel positions and moves the palm to the anterior facing (supinated) position. It helps to remember that supination is the motion you use when scooping up soup with a spoon (see Figure 4).
Dorsiflexion and Plantar Flexion
Dorsiflexion and plantar flexion are movements at the ankle joint, which is a hinge joint. Lifting the front of the foot, so that the top of the foot moves toward the anterior leg is dorsiflexion, while lifting the heel of the foot from the ground or pointing the toes downward is plantar flexion. These are the only movements available at the ankle joint (see Figure 4).
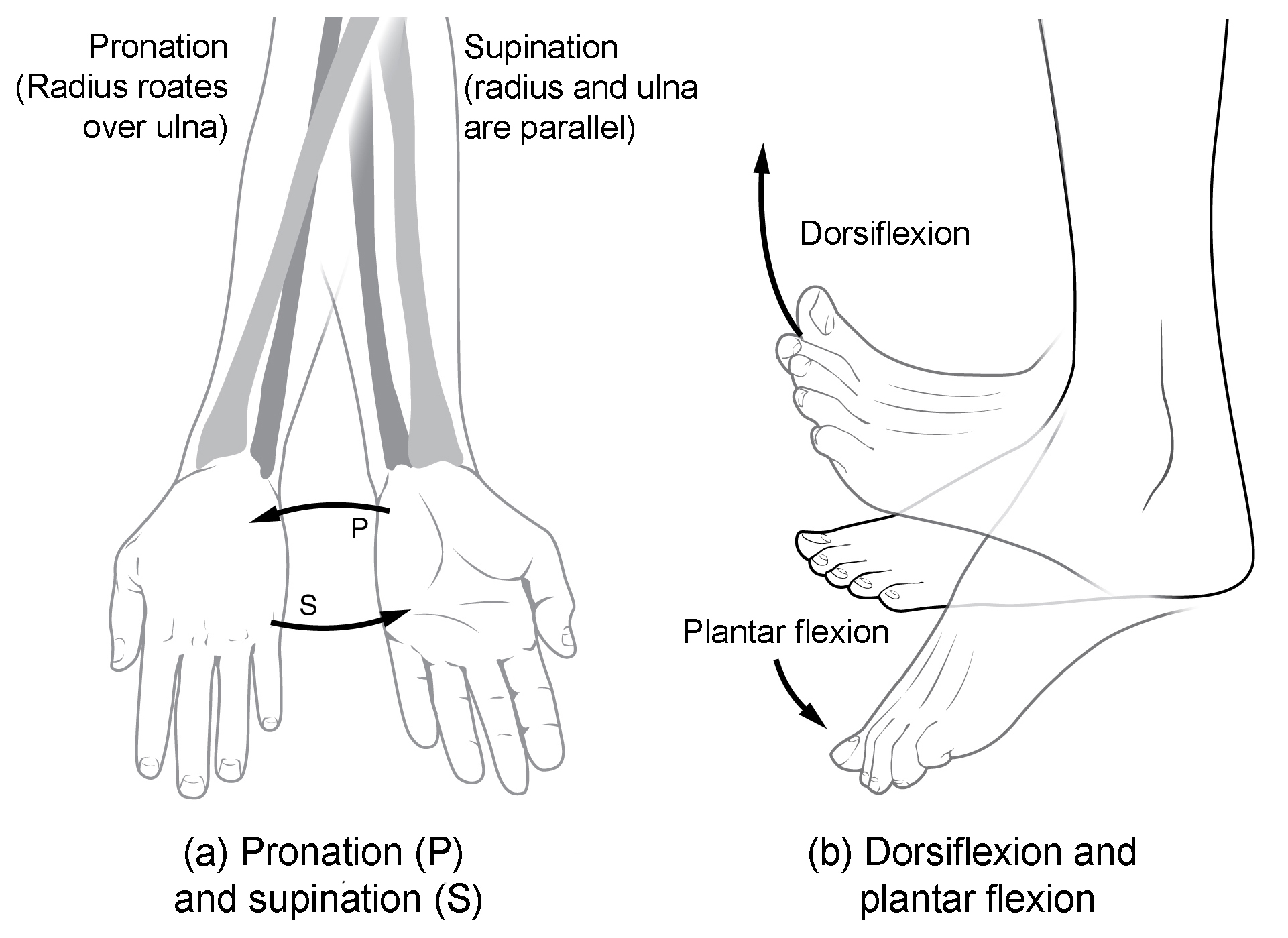
Figure 4. Supination and pronation. (a) Supination of the forearm turns the hand to the palm forward position in which the radius and ulna are parallel, while forearm pronation turns the hand to the palm backward position in which the radius crosses over the ulna to form an “X.” (b) Dorsiflexion of the foot at the ankle joint moves the top of the foot toward the leg, while plantar flexion lifts the heel and points the toes.
Inversion and Eversion
Inversion and eversion are complex movements that involve the multiple plane joints among the tarsal bones of the posterior foot (intertarsal joints) and thus are not motions that take place at the ankle joint. Inversion is the turning of the foot to angle the bottom of the foot toward the midline, while eversion turns the bottom of the foot away from the midline. The foot has a greater range of inversion than eversion motion. These are important motions that help to stabilize the foot when walking or running on an uneven surface and aid in the quick side-to-side changes in direction used during active sports such as basketball, racquetball, or soccer (see Figure 5).
Protraction and Retraction
Protraction and retraction are anterior-posterior movements of the scapula or mandible. Protraction of the scapula occurs when the shoulder is moved forward, as when pushing against something or throwing a ball. Retraction is the opposite motion, with the scapula being pulled posteriorly and medially, toward the vertebral column. For the mandible, protraction occurs when the lower jaw is pushed forward, to stick out the chin, while retraction pulls the lower jaw backward. (See Figure 5.)
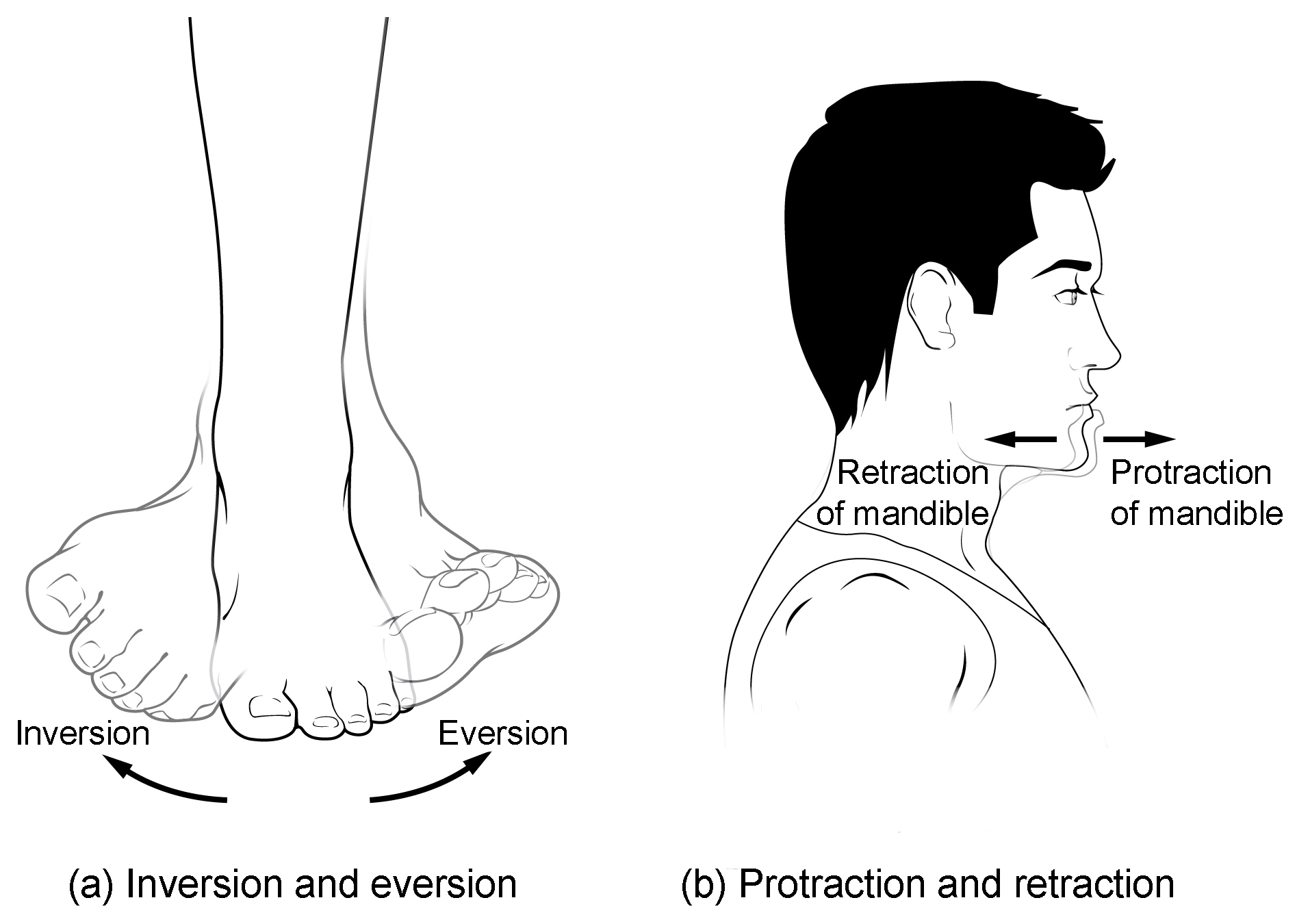
Figure 5. Inversion, eversion, protraction, and retraction. (a) Eversion of the foot moves the bottom (sole) of the foot away from the midline of the body, while foot inversion faces the sole toward the midline. (b) Protraction of the mandible pushes the chin forward, and retraction pulls the chin back.
Depression and Elevation
Depression and elevation are downward and upward movements of the scapula or mandible. The upward movement of the scapula and shoulder is elevation, while a downward movement is depression. These movements are used to shrug your shoulders. Similarly, elevation of the mandible is the upward movement of the lower jaw used to close the mouth or bite on something, and depression is the downward movement that produces opening of the mouth (see Figure 6).
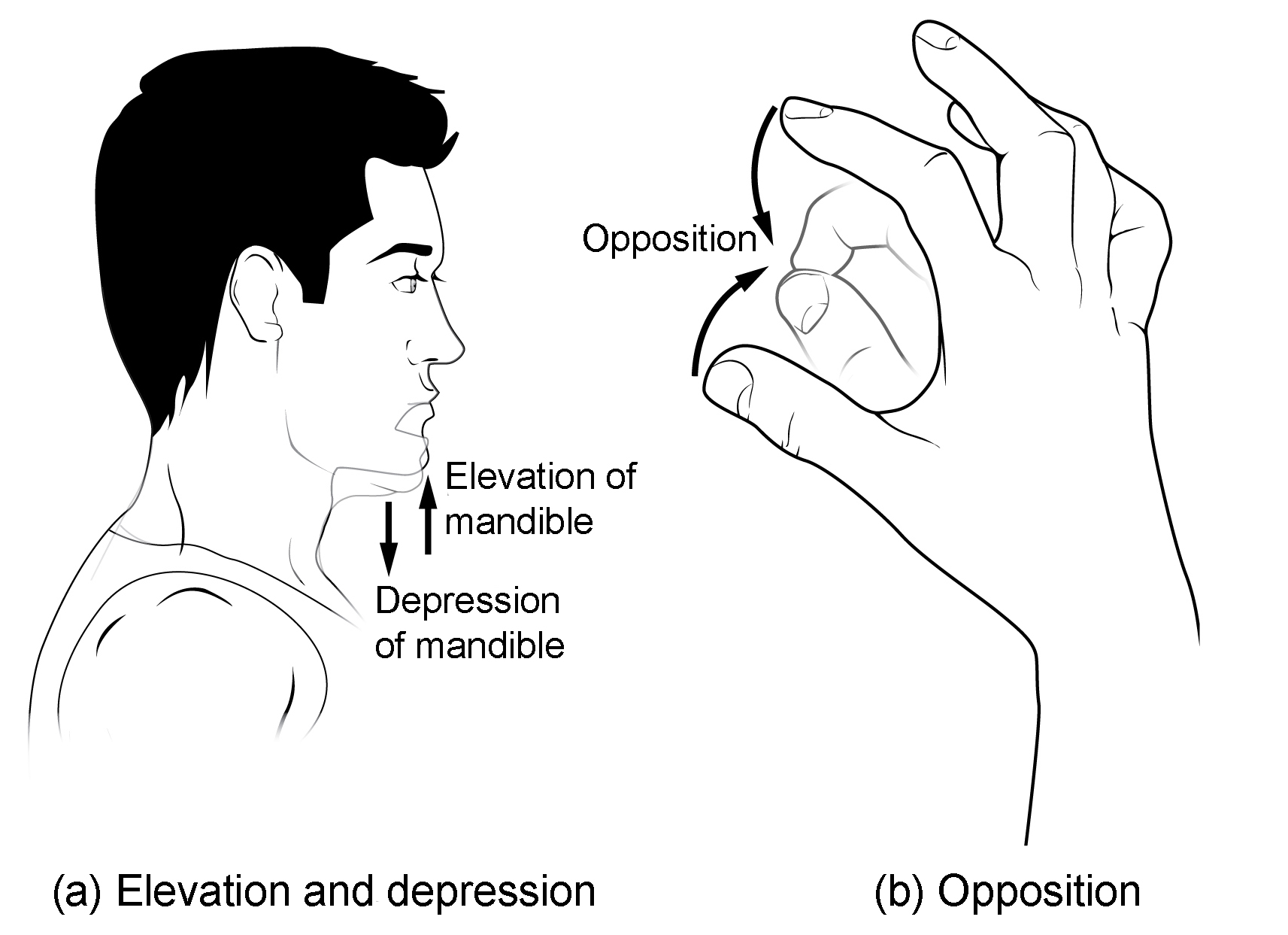
Figure 6. Depression, elevation, and opposition. (a) Depression of the mandible opens the mouth, while elevation closes it. (b) Opposition of the thumb brings the tip of the thumb into contact with the tip of the fingers of the same hand and reposition brings the thumb back next to the index finger.
Excursion is the side to side movement of the mandible. Lateral excursion moves the mandible away from the midline, toward either the right or left side. Medial excursion returns the mandible to its resting position at the midline.
Superior Rotation and Inferior Rotation
Superior and inferior rotation are movements of the scapula and are defined by the direction of movement of the glenoid cavity. These motions involve rotation of the scapula around a point inferior to the scapular spine and are produced by combinations of muscles acting on the scapula. During superior rotation , the glenoid cavity moves upward as the medial end of the scapular spine moves downward. This is a very important motion that contributes to upper limb abduction. Without superior rotation of the scapula, the greater tubercle of the humerus would hit the acromion of the scapula, thus preventing any abduction of the arm above shoulder height. Superior rotation of the scapula is thus required for full abduction of the upper limb. Superior rotation is also used without arm abduction when carrying a heavy load with your hand or on your shoulder. You can feel this rotation when you pick up a load, such as a heavy book bag and carry it on only one shoulder. To increase its weight-bearing support for the bag, the shoulder lifts as the scapula superiorly rotates. Inferior rotation occurs during limb adduction and involves the downward motion of the glenoid cavity with upward movement of the medial end of the scapular spine.
Opposition and Reposition
Opposition is the thumb movement that brings the tip of the thumb in contact with the tip of a finger. This movement is produced at the first carpometacarpal joint, which is a saddle joint formed between the trapezium carpal bone and the first metacarpal bone. Thumb opposition is produced by a combination of flexion and abduction of the thumb at this joint. Returning the thumb to its anatomical position next to the index finger is called reposition (see Figure 6).
- Chapter 9. Authored by : OpenStax College. Provided by : Rice University. Located at : http://cnx.org/contents/[email protected]@7.1. . Project : Anatomy & Physiology. License : CC BY: Attribution . License Terms : Download for free at http://cnx.org/content/col11496/latest/

- Topic Home / Introduction
Basic Mandibular Movements
- CR, RCP, and ICP
- The Slide and The IDC
- The Posselt Envelope
- 3D Animations
- Dynamic Occlusion
- Interferences
- Conform or Re-organise?
Practical procedures
- Facebow Transfer
- Recording ICP
- Recording Centric Relation
- Lateral and Protrusive Records
- Incisal Guidance Table
- Fabricating a lucia jig
Self Assessments
- Basic Principles
- Articulators
- Exam Pass Notes
- Record Your Reflections
- View all Module Notes
- Give Module Feedback
Occlusion Static Occlusion, Dynamic Occlusion and Guidance
Previous pages have looked at mandibular movements in the sagittal (forward-back) plane. We will now examine the effects of the mandible moving in lateral excursions.
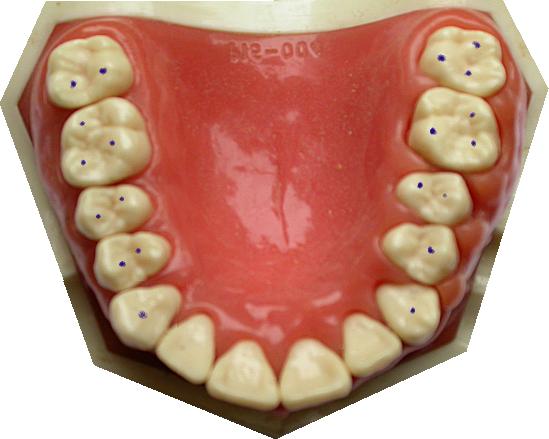
Static occlusion
Static occlusion is the study of contacts between the teeth when the jaw is not moving. The contacts are points (seen as dots when articulating paper is used). For example, close your teeth in your intercuspal position (i.e. where they meet best). Is your bite heaviest on your back teeth? Is it evenly spread over all the back teeth? Is there hardly any weight on the front teeth? Many dentists would describe that as the “ideal”. If you are providing a crown or filling for a patient like this, you would try not to disturb such an occlusion by leaving the restoration high (or low). Click the picture above to see the ink marks made with a static occlusion (intercuspal position in this case - the patient was asked to "tap together on the back teeth"). more… All these bite marks have been Photo-shopped onto the pictures. They are rarely this neat in real life! Top Tip: To get neater marks, smear the articulating paper with Vaseline first. This helps transfer the ink to the teeth.
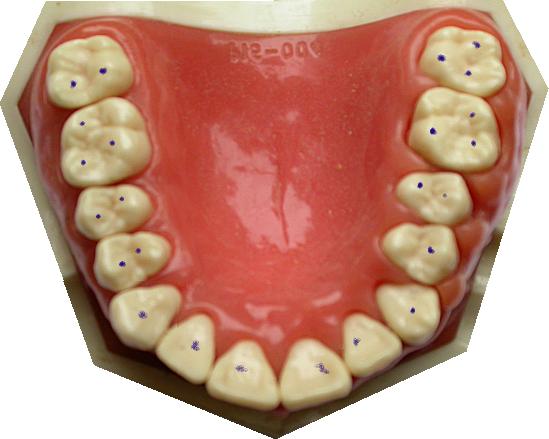
(No) Freedom In Centric
Bite on your back teeth. Can you slide a tiny way forwards before you bump into your anteriors? If so, you have "Freedom In Centric Occlusion" (also known as "Long Centric"). Remember - Centric Occlusion is another word for Intercuspal Position (ICP). The picture above shows no freedom in centric - ICP causes marks to appear on the incisors. Compare it with the picture at the page top, which has freedom in centric. Dynamic occlusion Dynamic occlusion is the study of the contacts that teeth make when the mandible is moving – contacts when the jaw moves sideways, forwards, backwards, or at an angle. The contacts are not points, they are lines. If you get a patient to grind their teeth in every direction on piece of articulating paper, you will see the lines formed by dynamic occlusion Although the jaw is moved by muscles, the contact lines depend on both the teeth positions and shapes (obviously), and the shape of the TMJ. These shapes GUIDE the occlusion. You may have heard the phrases posterior guidance and anterior guidance used when the mandible moves about in protrusive and lateral excursions Can you say what the terms mean? Check answer Posterior guidance is provided by the TMJ. As the condyle slides down the glenoid fossa, the angle of the mandible is changed depending how far the condyle is down the fossa’s slope. Anterior guidance is provided by the teeth. Not all of them, just the ones in contact during the movement. my score: 0 / 2 running total: 0 / 0 Reset Did you think posterior meant back teeth and anterior meant front teeth? Not here it doesn’t! Put your teeth together and slide all the way forwards: probably you can feel only your incisors “guiding”. Now imagine you had no upper front teeth or upper premolars at all, only molars on the top. In this case when you protrude, the “anterior guidance” is provided by molars. Some dentists prefer to use the phrase Anterior Determinant for teeth, and Posterior Determinant for the TMJs. This helps reduce the confusion. my score: 0 / running total: 0 / 0 Reset Working side and non-working side. Can you define these terms? Check answer If the mandible moves to the right , the right is the working side (and left is non-working). If the mandible moves to the left , the left is the working side (and right is non-working). my score: 0 / 2 running total: 0 / 0 Reset Types of anterior guidance in lateral excursions. Click the pictures to see typical ink marks associated with each type of guidance. Note the dots formed by intercuspal position (static), and the lines caused by a lateral excursion to the right.
These interferences are providing anterior guidance, but as the name suggests, not in a way that dentists approve of ! They are considered the least desirable types of anterior guidance.
Please take a moment to review these points, reflect on their significance and consider how they apply to your own experiences.
Tick to confirm (optional):
- Static occlusion refers to the contacts between teeth when the jaw is not moving, usually assessed in Intercuspal Position (ICP).
- Freedom in Centric Occlusion (Long Centric) allows slight forward movement before anterior teeth contact, while no freedom causes immediate anterior contact.
- Dynamic occlusion studies the contact lines formed when the jaw moves, influenced by both tooth and TMJ shapes.
- Anterior guidance is provided by teeth during movement, while posterior guidance is provided by the TMJ.
- Working side refers to the side the jaw moves towards, with the opposite side being the non-working side.
- Canine guidance occurs when only canines contact during lateral movement, considered ideal due to canines' ability to handle lateral forces.
- Group function involves multiple teeth sharing the load during lateral excursions, considered second best to canine guidance.
- Working side interference occurs when a single posterior tooth contacts first during lateral movement, and non-working side interference involves contact on the opposite side.
Membership Options
Dentaljuce offers a range of membership options…
Regular Membership
With enhanced CPD Certificates. Dentaljuce is brought to you by the award winning Masters team from the School of Dentistry, University of Birmingham, UK. All have won awards for web based learning and teaching and are recognised as leaders and innovators in this field, as well as being highly experienced clinical teachers. Full access to over 100 courses, no extras to pay.
£89.00 per year
Student Membership
No Certificates. With universities cutting down on traditional lectures, many students are currently having to rely more on online resources. If you don't need CPD Certificates, we are offering an amazing discount on your Dentaljuce personal membership fee. Special student price just £29 for 12 months individual membership.
£29.00 per year
© Dentaljuce 2024 | Terms & Conditions | Privacy Policy
Pocket Dentistry
Fastest clinical dentistry insight engine.
- Dental Hygiene
- Dental Materials
- Dental Nursing and Assisting
- Dental Technology
- Endodontics
- Esthetic Dentristry
- General Dentistry
- Implantology
- Operative Dentistry
- Oral and Maxillofacial Pathology
- Oral and Maxillofacial Radiology
- Oral and Maxillofacial Surgery
- Orthodontics
- Pedodontics
- Periodontics
- Prosthodontics
- Gold Member
- Terms and Condition
6: OCCLUSION
6.1 introduction to occlusion.
Working impression
Casting working model
Occlusion is the subject that is concerned with how the teeth and associated bones, joints and muscles function together.
The natural dentition
When you put your teeth together, the occlusal surfaces meet in the same position each time ( Figure 6.1.1 ). This position is called intercuspal position (ICP) and is used extensively in dentistry. ICP is a relationship between the maxilla and mandible when the teeth are in maximum intercuspation or maximum meshing. Other terms used for ICP are centric occlusion or habit bite.
Figure 6.1.1
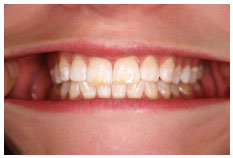
In the ICP, the occlusal load is distributed through the molars. You can feel this if you squeeze your teeth together very hard; there should be little or no pressure on your anterior teeth. The molars are well suited to distribute this load as the roots have a large surface area with which to transmit the load to the bone.
Figure 6.1.2
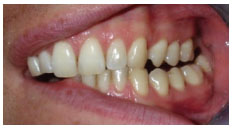
Figure 6.1.3
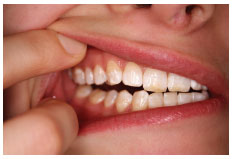
From ICP, move your mandible to the left with your teeth in contact. In most cases you will find that the canines are the only teeth contacting or working together ( Figure 6.1.2 ). In this excursion or movement from ICP, the left side is called the working side. The teeth on the right should not be contacting or not working ( Figure 6.1.3 ), which is why this side is termed the non-working side.
Of course when moving your mandible to the right from ICP, you should find the right canines contact, therefore the right is now the working side and the left teeth have space between them as this is the non-working side.
In these lateral excursions, only the canines contact and take the occlusal load, while the other teeth are separated. The canines are better suited than the posterior teeth to distribute these sideways forces for several reasons.
- Their shape is strong enough to resist force unlike molar teeth that have fissures causing weaknesses.
- They have long roots that prevent them from moving or tipping.
- They are further from the mandibular hinge (the temporomandibular joint (TMJ)) and therefore the muscles cannot exert such high forces.
- They are also more highly innervated or sensitive than other teeth and can easily detect light contact. This stimulus informs the brain, which in turn reduces the load on the tooth. You can try this for yourself, squeeze together in ICP and feel your masseter muscle on the angle of your mandible. Now move you jaw sideways such that only the canines on one side touch and squeeze in this position. You should feel that the brain has limited the activity of the muscle.
When the canine dictates the sideways movement of the mandible, the occlusal scheme is considered to be canine guided. This means that the canine is the only tooth guiding the sideways movement of the jaw.
Figure 6.1.4
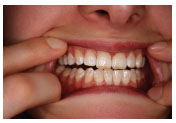
The anterior teeth act in the same way during forward movements of the mandible, dictating is pathway and causing the posterior teeth to separate ( Figure 6.1.4 ).
Figure 6.1.5
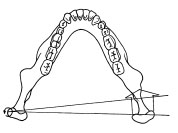
The two TMJs also dictate the mandibular movement at the posterior end. As the jaw moves to the left, the left or working side condyle rotates on an axis through the head as shown in Figure 6.1.5 . This working side condyle may also be termed the rotating condyle.
Figure 6.1.6
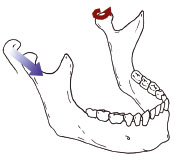
Figure 6.1.7
The right side condyle moves forwards, down the articular eminence (the bony slope of the joint) and towards the left at the same time. This non-working side condylar pathway is guided by the articular eminence ( Figure 6.1.6 ). Both condyles may translate (slide) forward in the protrusive movement of the jaw ( Figure 6.1.7 ).
Centric relation
The mandible can be related to the maxilla in one more relationship, this time determined by the position of the condyles rather than the teeth. The condyles need to be located in their optimal position at the top of the articular eminence ( Figure 6.1.8 ). Here the condyles can distribute any load through the bony structures of the skull rather than using muscles to hold the condyle on the slope of the articular eminence.
Figure 6.1.8
When the condyles are in this position the mandible can hinge open and closed by approximately 20 mm without any forward movement. This range of movement is called centric relation (CR) and again is used extensively in dentistry. You should assume that in an ideal situation ICP coincides with the CR arc of closing, therefore when the mandible is closed in CR, the teeth meet in ICP. This is the basic arrangement and function of the teeth in the idea/>
Share this:
- Click to share on Twitter (Opens in new window)
- Click to share on Facebook (Opens in new window)
Related posts:
- 7: SHADE AND COLOUR
- 1: INTRODUCTION
- 5: ORTHODONTICS
- 3: PARTIAL PROSTHETICS
- 2: COMPLETE PROSTHETICS
- 3: Retaining Abutment Screws
Stay updated, free dental videos. Join our Telegram channel
Comments are closed for this page.

VIDEdental - Online dental courses


- Dr Hirschinger
- Office Info
- Facial Pain
- Sleep Apnea
- Testimonials
TMJ Muscle Range of Motion Measurements

The notch fits into the midline of the lower central incisors.
TMJ Muscle Range of Motion Measurements A patient’s range of motion measurements should be recorded as part of every new patient exam. This adds anywhere from 20 seconds to a new patient exam if you only record the three opening movements, which are “comfort, active, and passive.” If you measure right and left lateral and protrusive movements, you might be adding 60 seconds to your exam time. Yes, it’s that quick.
Normal Range of Motion Opening : 40 to 60 mm. If a patient opens to 55 mm with either the comfort or active opening, there is no need to obtain the Passive opening. Additionally, do not have a patient open past 60 mm and do not push a patient open past 60 mm. If they can open to 60 mm or you can push them to 60 mm, then they have an excellent opening range of motion. Lateral : 8 to 12 mm Protrusive : 8 to 12 mm
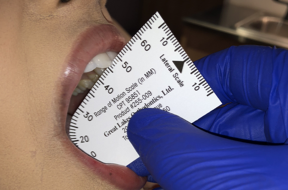
Note in your chart if any of the movements cause pain and if any of the movements replicate their jaw pain. If a movement does cause pain, have the patient point with one finger where the pain occurs.
If the patient ever has a future problem with opening and/or moving their jaw, you now have a baseline of what their normal movements are. Do you think it is worth spending up to one additional minute of your time to record these measurements? You will if you ever have a patient who has a problem especially if they think you created it with an injection or by keeping them open too long.
As always, I’m happy to help any patient who is experiencing jaw pain or any facial pain including migraines, trigeminal neuralgia, sleep apnea or snoring issues at my office in Beverly Hills. Please let me know how I can help or if you have any questions.
2 Responses
i would like to order your measuring device. how do I do this.
carl patrnchak, pt, co
I just noticed your request so I apologize for the delay. The Range of Motion scale is not something I created. I buy mine from https://www.greatlakesortho.com/commerce/detail/?nPID=1352&CFID=3860914&CFTOKEN=bc32584062189817-8A7DC824-5056-9F7B-7994252789E82D7B
Leave a Reply
You can use these tags: <a href="" title=""> <abbr title=""> <acronym title=""> <b> <blockquote cite=""> <cite> <code> <del datetime=""> <em> <i> <q cite=""> <s> <strike> <strong>
Registered Nurse RN
Registered Nurse, Free Care Plans, Free NCLEX Review, Nurse Salary, and much more. Join the nursing revolution.
Body Movement Terms – Anatomy Body Planes of Motions
In this anatomy lesson, I’m going to cover all of the major body movement terms for anatomy (also called the planes of motion ) that can occur at the synovial joints. You’ll come across these in your anatomy or kinesiology courses, and if you pursue a career in healthcare, you’ll use these terms during documentation or patient assessments.
After you review these notes and the corresponding video, you can take a comprehensive quiz on anatomy body movement terms .
Categories of Body Movement Terms in Anatomy
There are four major categories of body movements that can occur at the synovial joints:
- Gliding movement
- Angular movements
- Rotational movements
- Special movements
Gliding Movement in Anatomy
What is gliding ? Gliding occurs when the surfaces of bones slide past one another in a linear direction, but without significant rotary or angular movement.
An example of this movement is moving your hand back and forth (left to right) in a waving motion, which causes gliding to occur at the joints of the carpals ( wrist bones ). When you move your hand back and forth in a waving motion, it can help you remember that gliding joint movements primarily take place in the carpals of the wrist and the tarsals of the ankle.

However, gliding can also occur in the other plane joints (also called planar joints) of the body. Just as airplanes glide through the air, the plane joints of the body allow a gliding motion.
Other plane joints that allow gliding include the sacroiliac joint of the pelvis , the acromioclavicular joint of the shoulder, the femoropatellar joint , tibiofibular joint , sternocostal joints for ribs 2-7, vertebrocostal joints , and the intervertebral joints of the spine (at the articular processes).
Angular Movements in Anatomy
The next category of body movement terms consists of the angular movements, which consist of the following movements:
- Flexion and Extension
- Abduction and Adduction
- Circumduction
Flexion and Extension in Anatomy
Because flexion and extension are angular movements, I find it really helpful to visualize an angle during the actual movement. Flexion decreases the angle between two structures or joints as they bend or move closer together, whereas extension increases the angle between them as they straighten and move apart.
Elbow Flexion and Extension
Elbow flexion (also called forearm flexion ) occurs when the angle between the forearm and arm decreases, allowing the ulna of the forearm to move closer to the humerus bone of the arm.
In contrast, elbow extension ( forearm extension ) occurs when the forearm moves away from the arm, increasing the angle between those bones.

Shoulder Flexion and Extension
Shoulder flexion , also called arm flexion , occurs when the angle at the humerus of the arm and the scapula decreases as the arms move anteriorly. In contrast, shoulder extension (or arm extension ) occurs when the angle at the humerus of the arm and the scapula increases, causing the arm to move posteriorly. The joint here allows movement past the anatomical position. Some anatomists call arm movement beyond the anatomical position extension , whereas some call it hyperextension .
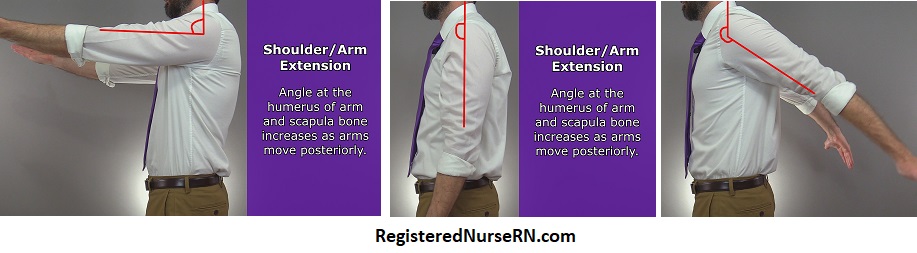
Wrist Flexion and Extension
Wrist flexion (also called hand flexion ) occurs when the angle between the palm of the hand and the anterior surface of the forearm decreases, while wrist extension (or hand extension ) is moving the palm of the hand away from the anterior surface of the forearm, hence the angle increases. This is another joint that can continue to move past the anatomical position in a posterior direction, which some anatomists call hyperextension.

Finger Flexion and Extension
Finger flexion occurs when the angle between the fingers and the palm decreases, as the fingers move toward the palm. When the angle between the fingers and the palm increases, finger extension occurs.

Flexion and extension also occur with the interphalangeal joints of the fingers (digits 2-5), including the distal interphalangeal joint (dip) and proximal interphalangeal joint (pip).

Thumb Flexion and Extension
The thumb (pollex) can confuse people because thumb flexion and extension occur in the frontal plane , which is a different direction than flexion of the fingers, which occurred in the sagittal plane. Thumb flexion moves the thumb toward the pinky finger, whereas extension moves the thumb away from the pinky finger. Think of your palm as a windshield and your thumb as the windshield wiper for this movement.

Flexion and extension can also occur at the interphalangeal joint of the thumb.

Hip Flexion and Extension
Hip flexion (or thigh flexion ) occurs when the angle between the femur of the thigh and hipbone decreases as the thigh moves anteriorly (forward). Hip extension ( thigh extension ) occurs when the angle between the femur and the hip bone increases, as the hip joint straightens. This joint also allows posterior movement past the anatomical position, which some anatomists call hyperextension.
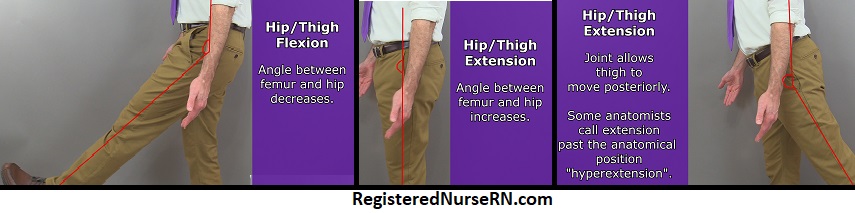
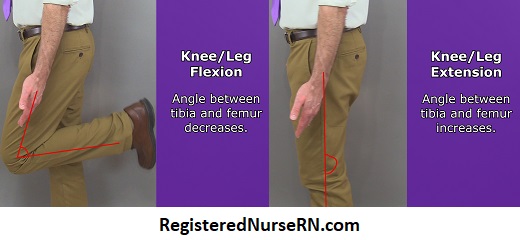
Knee Flexion and Extension
Knee flexion ( leg flexion ) occurs when the tibia bone moves toward the femur, causing the angle to decrease between those two structures. Knee extension (or leg extension ) occurs as the angle between the leg bones increases, causing the leg to straighten.
Toe Flexion and Extension
Like the fingers, toe flexion and extension can also occur. Toe flexion involves bending the toes toward the sole of the foot, decreasing the angle between these two structures, while toe extension involves increasing the angle and straightening the toes.

Note : instead of using flexion and extension for the movement of the foot at the ankle joint, anatomists prefer to use the terms plantarflexion and dorsiflexion .
Neck Flexion and Extension
Neck flexion occurs as the angle between the head and the trunk of the body decreases as those two structures move closer together, whereas neck extension occurs as the head moves away from the trunk of the body, thus increasing the angle. The neck is another structure that can continue posteriorly, beyond the anatomical position, which some anatomists call hyperextension of the neck .

Vertebral Column Flexion and Extension
Vertebral column flexion at the trunk, (spine flexion) occurs when the angle between the trunk and the hip joint decreases. Vertebral column (spine) extension at the trunk occurs as the spine straightens and the angle between the hip joint and spine increases.

By the way, you might have noticed that most of these movements so far are occurring within (or parallel to) the sagittal plane. However, just like the thumb, flexion can also occur in the frontal (coronal) plane for the vertebral column. For example, if you bend the spine to the left or right, that’s called lateral flexion , and movement back toward the anatomical position is called lateral extension .

Note: you might want to watch our other lecture if you are unfamiliar with the different body planes .
Hyperextension
Finally, when extension of a structure moves beyond a certain point, anatomists call it hyperextension. However, anatomists differ on what constitutes hyperextension when it comes to body movement terms.

For example, some anatomists say that when the arm , neck , wrist , or thigh moves past the anatomical position in a posterior motion, it becomes hyperextension . Other anatomists only consider these movements hyperextension if the movement exceeds the normal range of motion permitted by the joint. For test-taking purposes, follow your anatomy teacher’s definition!
Abduction and Adduction in Anatomy
Unlike flexion and extension movements, which mostly take place within the sagittal plane , you’ll notice that abduction and adduction motions mostly take place within the frontal, or coronal, plane. However, the thumb is a notable exception to this rule, as it moves within the sagittal plane during abduction and adduction when in the anatomical position .
What is Abduction?
Abduction (think: ABDUCT ion) is the movement of a structure away from its midline reference point. Let the name help you out. What does “abduct” mean? When you hear on the news that a man was abducted, you know it means that someone took him away . That’s exactly what’s going on with this movement. The structure is being moved away from its midline reference point.
What is Adduction?
Adduction (think: ADD uction) occurs as the structure is ADDED back toward its midline reference point.
Let’s take a look at examples of abduction and adduction on the body.
Arm Abduction and Adduction
During arm abduction (also called shoulder abduction), the arms move away from the body’s midline. During arm adduction (or shoulder adduction), you ADD them right back toward the midline.

Finger Abduction and Adduction
Finger abduction occurs when the fingers move away from the midline reference of the hand, whereas finger adduction occurs when you add them back toward the hand’s midline reference.
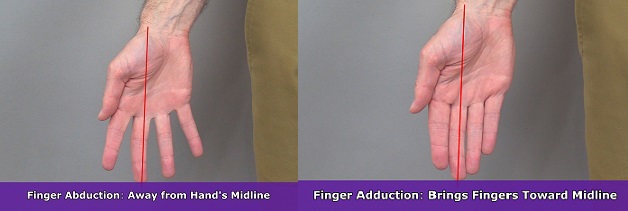
When the middle finger (3rd digit), which serves as the midline reference of the hand, deviates to the away from the body, it’s called lateral abduction . When it deviates toward the body, it’s called medial abduction .

Thumb Abduction and Adduction
The thumb (pollex) is different from the fingers. Abduction of the thumb has it moving within the sagittal plane, in an anterior motion. Adduction of the thumb has it added back to the hand.

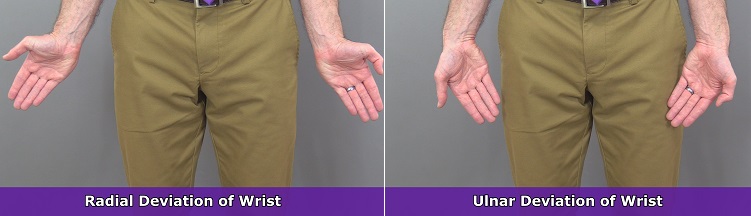
Wrist Abduction and Adduction (Ulnar Deviation & Radial Deviation)
When determining abduction and adduction of the wrist, I find that it helps to stand in the anatomical position. Abduction of the wrist has it moving away from the body’s midline, in the same direction as arm abduction. Adduction of the wrist has it going in the opposite direction, toward the body’s midline.

These movements are also referred to as radial deviation and ulnar deviation . Remember, the radius is on the thumb side, which where you check the radial pulse. So radial deviation is movement on the radial side, whereas ulnar deviation occurs on the opposite side.
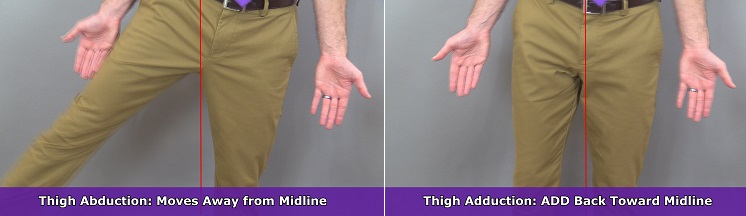
Thigh Abduction and Adduction
During thigh abduction (also called hip abduction or leg abduction), the lower limb moves away from the body’s midline. During adduction of the thigh , you ADD the lower limb right back toward the body’s midline.
Toes Abduction and Adduction
When the toes move away from the midline of the foot, toe abduction occurs. Toe adduction adds them right back together.

Just like with the hand, devation of the 2nd toe away from the body’s midline is called lateral abduction , whereas movement toward the midline is called medial abduction .

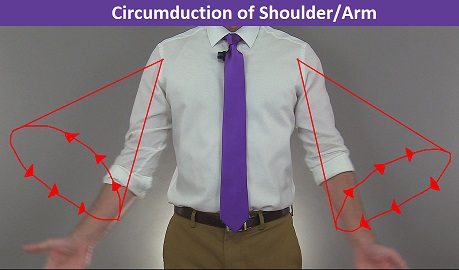
Circumduction in Anatomy
The final body movement term in this category is circumduction , which is an angular movement that blends the motions of flexion , abduction , extension, and adduction to create a circular or conical motion of the attached structure.
The word circ umduction starts with the same letters as the word “ circ le,” so that will tip you off that this movement creates a circular, or conical, movement in the structure extending beyond the joint.
Circumduction Movement Demonstrated
Because circumduction is a combined movement, I find it helpful to think about the individual movements in slow motion. Looking at the shoulder joint, I’ll begin with arm flexion and then arm abduction. Next is arm extension , followed by arm adduction . When you combine those movements into one smooth motion, you can see how it forms a cone or circle.
The mnemonic “FABEA” might help you remember the order:
You could also reverse that order, but the movements have to alternate in a similar succession to create the circular motion that characterizes the circumduction movement.
Joints Capable of Circumduction
Where can circumduction occur on the body? Because it requires the motions of flexion, extension, abduction, and adduction, the joint will generally have to be capable of all four of those sequential movements. Below are examples of joints/structures that can perform the circumduction movement.
Circumduction of the Hip Joint (Thigh)

Circumduction of the Shoulder Joint (Arm)
Circumduction of the Wrist Joint (Hand)
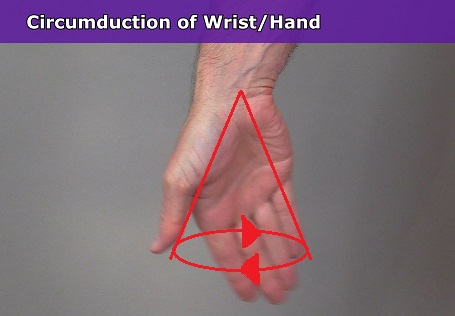
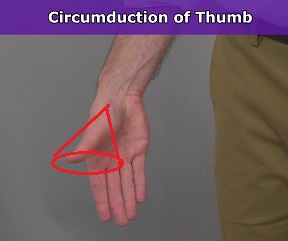
Circumduction of the Thumb (Pollex)
Circumduction of the Fingers

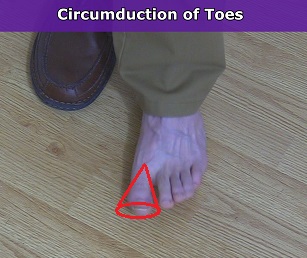
Circumduction of the Toes
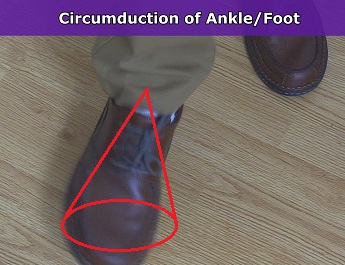
Circumduction of the Ankle Joint (Foot)
Circumduction of the Head

Rotation in Anatomy
The next category of the body planes of motion is rotation , which is a body movement term that describes a bone moving around a central axis.
Rotation Body Movement Term in Anatomy
When I think of the rotation body movement , I like to picture a screw turning to either the right or left, as that is similar to the rotation movement that can occur in the body.
Rotation can occur at the head/neck, vertebral column, and the ball-and-socket joints of the upper and lower limbs (shoulder joint and hip joint). Let’s take a look at these movements, starting with the head.
Head and Neck Rotation
The head can rotate laterally to either the left or right, thanks to the pivot joint between vertebrae C1 (atlas) and C2 (axis). Moving the head back toward the anatomical position is medial rotation of the head.

Trunk Rotation
The vertebral column can also rotate laterally to either the left or right. Returning the trunk back toward the anatomical position is medial rotation of the trunk.

Arm Rotation (Medial and Lateral)
The ball-and-socket joint of the shoulder allows the humerus of the arm to rotate laterally, or away from the body’s midline , which is also called external rotation. It can also rotate medially, or toward the body’s midline, which is also called internal rotation.

Thigh/Leg Rotation (Medial and Lateral)
The ball-and-socket joint of the hip allows rotation of the thigh’s femur . Like the humerus, it can rotate laterally, or away from the body’s midline, which is also called external rotation. It can also rotate medially, or toward the body’s midline, creating an internal rotation movement.

Tip for Medial vs Lateral Limb Rotation
Be sure to focus on the anterior surface of the femur or humerus when you do this movement, because that’s the focal point for determining medial vs lateral rotation.

Special Body Movement Terms in Anatomy
The final category of body movement terms include the “special movements.” These movements don’t fall neatly into the categories I’ve already listed, so they are placed in their own unique category. The special movements involve the following:
Supination and Pronation
- Dorsiflexion and Plantarflexion (also spelled plantar flexion)
Inversion and Eversion
Elevation and depression.
- Protraction and Retraction
- Protrusion, Retrusion, and Excursion
- Opposition and Reposition
Supination and pronation are special movements involving rotation of the forearm .
Supination of Forearm and Hand
During supination , the distal end of the radius bone rotates over the ulna bone in a lateral direction . Lateral rotation means it is rotating away from the body’s midline.
I like to watch the thumb during this movement, because it is on the same side as the radius (hence, the radial pulse is located below the thumb). When the thumb is rotating away from the body’s midline, supination is occurring.


Pronation of Forearm and Hand
In contrast, pronation is the opposite movement: the distal end of the radius rotates over the ulna medially, and the two bones cross. Medial rotation is toward the body’s midline. So when the thumbs point toward the middle of the body, you know that pronation has occurred.

Palm Orientation During Supination and Pronation
You can also look at the orientation of the palms. During supination, the palms will face anteriorly (forward), which is their natural orientation in the anatomical position. However, if you flex the elbow about 90 degrees, the palms would then be facing up (superiorly).

Pronation has the palms facing the opposite direction: posteriorly (toward the back) when in the anatomical position, or down (inferiorly) when the elbow flexes to around 90 degrees. This is another reason why I like to look at the thumbs during this movement. Thumbs will point away from the body’s midline during supination, and toward the body during pronation, regardless of how the elbow is flexed.

Supination vs Pronation Mnemonic
Here’s a simple mnemonic (memory trick) to help you remember pronation vs supination special movements:
At the grocery store, you pro nate to pick up your pro duce, and you sup inate to eat it for sup per.

Also, if you want to take your vitamins, you pro nate to p our, and you sup inate to take your sup plements.
Plantarflexion and Dorsiflexion
In this continued series on body movements of anatomy, I’m going to demonstrate dorsiflexion and plantarflexion (or plantar flexion), which are special movements involving the foot and ankle joint.
Dorsiflexion vs Plantarflexion
To help you understand this special movement, let’s break down the words.
Dorsal Side of the Foot (Dorsum)
Dorsal refers to the back (or upper) side of something. In my video on body cavities and membranes , I used the example of a dorsal fin of a dolphin to help you remember that dorsal refers to the backside of a surface. Your toenails are on the dorsal side of the foot, because they are on the back (or upper) side of it.
Plantar Side of Foot (Sole)
In contrast, plantar refers to the sole (or bottom) of the foot. If you’ve ever had a plantar wart, then you’ve had a wart on the sole of your foot (ouch!).
Flexion Meaning
Flexion refers to the movement that decreases the angle between two surfaces or joints, usually within the sagittal plane of the body. Now, let’s put all these words together, and you’ll be able to remember the difference between plantarflexion vs dorsiflexion .
Dorsiflexion Example

During dorsiflexion , the back (upper) side of the foot moves toward the shin, decreasing the angle between these two surfaces, leaving the toes pointing closer toward your head. When you try to walk on your heels only, you dorsiflex the foot.
Plantar Flexion (Plantarflexion) Example

During plantar flexion , the sole of the foot angles downward toward the calf, decreasing the angle between those two surfaces, leaving the toes pointing farther away from the body. When you perform calf raises in the gym or walk on your tip toes, you plantar flex the foot. If you need help remember the direction of this movement, just remember this phrase: “Plantarflexion helps you stand on your toes and walk in any direction!”
Next, I’m going to demonstrate inversion and eversion , which are special movements that cause the foot to move relative to the body’s midline.
Inversion of the Foot
During inversion , the bottom of the foot (sole) turns so that it faces toward the body’s midline, in a medial orientation. Inversion starts with the word “in,” so that’s the dead giveaway that the sole is pointing in wardly (medially).
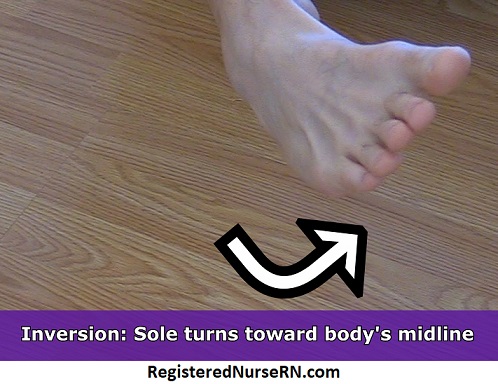
Eversion of the Foot
During eversion , the opposition motion occurs: the bottom of the foot turns so that it faces away from the body’s midline (laterally). The word “evert” literally means to “turn outward,” which is exactly what happens during eversion!

Now lets’s examine elevation and depression, which are special body movement terms that describe motion in a superior (up) or inferior (down) direction.
Elevation in Anatomy
Elevation refers to movement of a body part in a superior direction, or moving upward. When you walk into a hotel lobby, you have to get on the elevator to go up, right? We’d also say that a mountain has a peak “elevation” of 20,000 feet. Therefore, the term is pretty self-explanatory: elevation has a structure moving up, or superiorly.
Depression in Anatomy
Depression refers to movement of a body part in an inferior direction, or moving downward. When you are depressed, you feel down in the dumps, right? Therefore, depression is easy to remember as movement in an inferior, or downward direction.
Elevation and Depression in Anatomy
In anatomy, elevation and depression most commonly describe movements of the mandible (lower jaw) or scapulae (shoulder blades) within the frontal plane . When you move your lower jaw (mandible) in a downward direction, depression occurs. When you move your mandible upward, elevation occurs.

Similarly, when you move your scapulae up, elevation of the shoulder girdle occurs. When you move them back down, depression of the shoulder girdle occurs.

Protraction and Retraction in Anatomy
Now let’s discuss protraction and retraction , which are special body movements in anatomy that most commonly involve the scapulae (shoulder blades).
Protraction Movement
Protraction moves the scapula forward (anteriorly) and toward the side of the body (laterally) in an anterolateral direction.

Retraction Movement
Retraction is the opposite movement. It causes the shoulder blades to move back (posteriorly) and toward the body’s midline (medially). This movement is known as a posteromedial movement.

Protraction and Retraction Mnemonic
Here’s a simple way to remember protraction and retraction body movements in anatomy:
- You Retract when you Reach Back !
- You P unch to P rotract! In fact, the serratus anterior muscles assist with protraction of the scapulae, and they even call this muscle the boxer’s muscle for that very reason.
Protrusion, Retrusion, and Excursion in Anatomy
In this anatomy lesson, I’m going to demonstrate protrusion, retrusion, and excursion , which are special body movement terms in anatomy that refer to forward (anterior), backward (posterior), or side to side movements.
Protrusion in Anatomy
Protrusion refers to the movement of a structure in an anterior (forward) direction. In fact, the word protrude means “projecting something forward.”
I call protrusion the kissing movement because it occurs when you pucker your lips like you’re going to give someone a kiss or stick out your tongue. Moving the mandible (lower jaw) forward is also an example of protrusion.

Retrusion in Anatomy
Retrusion is the opposite of protrusion. It refers to the movement of a structure in a posterior, or backward, direction. Putting your tongue back in your mouth, moving the lips back, or moving the mandible back are all examples of retrusion in anatomy.
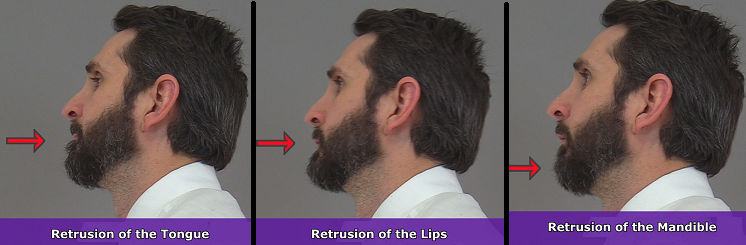
Excursion in Anatomy
Finally, we have excursion , which refers to the side-to-side movement of the lower jaw (mandible). If you’ve ever heard of a character named Ernest P. Worrell, then you’ve definitely seen the excursion movement. He’s the character in those movies such as Ernest Goes to Camp, Ernest Goes to Jail, etc. When Ernest saw something nasty, he’d move his jaw back and forth and say, “Ewwww.”

Excursion can occur in either direction, and anatomists use directional terms to specify the type of excursion. When the mandible moves to either the left or right, it’s moving away from the body’s midline, so it’s called lateral excursion . When the mandible moves closer to the midline of the body, it’s called medial excursion .

Protrusion and Retrusion vs Protraction and Retraction
What about protraction and retraction ? Some anatomy textbooks will refer to the forward movement of the mandible, lips, or tongue as protraction (instead of protrusion), and the backward (posterior) movement will be called retraction (instead of retrusion). The terms are sometimes used interchangeably, so use whatever method your anatomy professor suggests (they give you the grade, not me!).
However, some anatomists today use protraction and retraction to refer almost exclusively to the scapulae, as it is a combined movement (protraction is anterolateral, and retraction is posteromedial). In contrast, protrusion and retrusion are more of an anterior/posterior movement. Then again, some anatomists prefer not to use protraction and retraction at all, even when describing shoulder blade movement.
Opposition and Reposition of the Thumb: Anatomy
Finally, I’ll demonstrate opposition and reposition , which are special movements involving the thumb.
The thumb, also known as the pollex or digit one, articulates (forms a joint) with the trapezium bone of the wrist (carpus) via a saddle joint, which is a type of synovial joint featuring interlocking convex and concave surfaces. They call it a saddle joint because, well, it kinda looks like a saddle (yee-haw, cowboy!).
Thanks to this saddle joint, the thumb can perform circumduction, flexion and extension, abduction and adduction, as well as special movements called opposition and reposition .
Opposition of the Thumb
Opposition of the thumb occurs when the tip of the thumb comes to meet (and oppose) the tip of another finger from the same hand. A super easy way to remember this is that you’ve probably heard someone say that humans have opposable thumbs. Oppos ition is the special movement of our oppos able thumbs.

In fact, think about this: when the opposition movement occurs, what happens? In the picture above, did you notice how the thumb and finger created a shape similar to the letter ‘O’? The ‘O’ stands for opposition! Now you can easily remember this motion of our opposable thumbs!
Reposition of the Thumb
Reposition is the opposite action of opposition. During reposition, the thumb and finger return to their original position.

Free Quiz and More Anatomy Videos
Take a free comprehensive quiz on body movement terms to test your knowledge, or review our anatomy body movement terms video . In addition, you might want to watch our anatomy and physiology lectures on YouTube, or check our anatomy and physiology notes .
Please Share:
- Click to print (Opens in new window)
- Click to share on Facebook (Opens in new window)
- Click to share on Twitter (Opens in new window)
- Click to share on Pinterest (Opens in new window)
- Click to share on Reddit (Opens in new window)
- Click to share on LinkedIn (Opens in new window)
- Click to share on WhatsApp (Opens in new window)
- Click to share on Pocket (Opens in new window)
- Click to share on Telegram (Opens in new window)
Disclosure and Privacy Policy
Important links, follow us on social media.
- Facebook Nursing
- Instagram Nursing
- TikTok Nurse
- Twitter Nursing
- YouTube Nursing
Copyright Notice
- Remote Access
- Save figures into PowerPoint
- Download tables as PDFs

Chapter 13: The Temporomandibular Joint
- Download Chapter PDF
Disclaimer: These citations have been automatically generated based on the information we have and it may not be 100% accurate. Please consult the latest official manual style if you have any questions regarding the format accuracy.
Download citation file:
- Search Book
Jump to a Section
Structure and function.
- RESEARCH FINDINGS
- Full Chapter
- Supplementary Content
Temporomandibular Joint
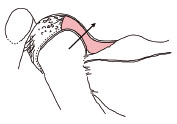
The temporomandibular joint (TMJ) is the articulation between the mandible, the articular disc, and the temporal bone of the skull ( Fig. 13.1A , B ). The disc divides the joint into two distinct parts, which are referred to as the upper and lower joints. The larger upper joint is formed by the convex articular eminence, concave mandibular fossa of the temporal bone, and the superior surface of the disc. The lower joint consists of the convex surface of the mandibular condyle and the concave inferior surface of the disc. 1 , 2 , 3 The articular disc helps the convex mandible conform to the convex articular surface of the temporal bone. 2
FIGURE 13.1
A: Lateral view of the skull showing the temporomandibular joint (TMJ) and surrounding structures. B: A lateral view of the TMJ showing the articular disc and a portion of the joint capsule.

The TMJ capsule is described as being thin and loose above the disc but taut below the disc in the lower joint. Short capsular fibers surround the joint and extend between the mandibular condyle and the articular disc and between the disc and the temporal eminence. 3 Longer capsular fibers extend from the temporal bone to the mandible.
The primary ligament associated with the TMJ is the temporomandibular ligament. The stylomandibular and the sphenomandibular ligaments ( Fig. 13.2 ) are considered to be accessory ligaments. 4 , 5 The muscles associated with the TMJ are the medial and lateral pterygoids, temporalis, masseter, digastric, stylohyoid, mylohyoid, and geniohyoid.
FIGURE 13.2
A: A lateral view of the temporomandibular joint showing the oblique fibers of the temporomandibular ligament and the stylomandibular and sphenomandibular ligaments. B: A medial view of the temporomandibular joint showing the medial portion of the joint capsule and the stylomandibular and sphenomandibular ligaments.

Osteokinematics
The upper joint is an amphiarthrodial gliding joint, and the lower joint is a hinge joint. The TMJ as a whole allows motions in three planes around three axes. All of the motions except mouth closing begin from the resting position of the joint in which the teeth are slightly separated (freeway space). 3 , 6 The amount of freeway space, which usually varies from 2 mm to 4 mm, allows free anterior, posterior, and lateral movement of the mandible.
The functional motions permitted are mandibular depression (mouth opening), mandibular elevation (mouth closing), protrusion (anterior translation; Fig. 13.3 ) and retrusion (posterior translation; Fig. 13.4 ), and right and left lateral excursion or laterotrusion (lateral deviation; Fig. 13.5 ). Maximal contact of the teeth in mouth closing is called centric occlusion.
Pop-up div Successfully Displayed
This div only appears when the trigger link is hovered over. Otherwise it is hidden from view.
Please Wait

TMJ Movements
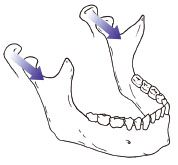
Normal movements of the jaw during function, such as chewing, are known as excursions. There are two lateral excursions ( left and right ) and the forward excursion, known as protrusion, the reversal of which is retrusion.
When the jaw is moved into protrusion, the lower incisors or front teeth are moved so that they first come edge to edge with the upper incisors, and then move past them, producing a temporary underbite. This is accomplished by sliding of the condyle down the articular eminance ( in the upper portion of the TMJ ) without any more than the slightest amount of rotation taking place ( in the lower portion of the TMJ ), other than that necessary to allow the lower incisors to come in front of the upper incisors without running into them.
During chewing, the jaw moves in a specific manner as delineated by the two TMJs. The side of the mandible that moves sideways is referred to as either the working or rotating side, while the other side is referred to as either the balancing or orbiting side. The latter terms, although a bit outdated, are actually more precise, as they define the sides by the movements of the respective condyles. When the jaw is moved into a lateral excursion, the working side condyle ( the condyle on the side of the jaw that moves outwards ) only performs rotation ( in the horizontal plane ), while the balancing side condyle performs translation. During actual functional chewing, when the teeth are not only moved side to side, but also up and down when biting of the teeth is incorporated as well, rotation ( in a vertical plane ) also plays a part in both condyles.
The jaw is moved primary by four muscles: the masseter, medial pterygoid, lateral pterygoid and the temporalis. These four muscles work in different groups to move the mandible in different directions. Contraction of the lateral pterygoid acts to pull the disc and condyle forward; thus, the action of this muscle serves to open the mouth. The other three muscles close the mouth; the masseter and the medial pterygoid by pulling up the angle of the mandible and the temporalis by pulling up on the coronoid process.
When the mouth opens, two distinct motions occur at the joint. The first motion is ROTATION around a horizontal axis through the condylar heads. The second motion is TRANSLATION. The condyle and meniscus move together interiorly beneath the articular eminence.
In the closed mouth position, the thick posterior band of the meniscus lies immediately above the condyle. As the condyle translates forward, the thinner intermediate zone of the meniscus becomes the articulating surface between the condyle and the articular eminence. When the mouth is fully open, the condyle may lie beneath the anterior band of the meniscus.
Musculoskeletal Key
Fastest musculoskeletal insight engine.
- MANUAL THERAPIST
- MUSCULOSKELETAL MEDICINE
- PHYSICAL MEDICINE & REHABILITATION
- RHEUMATOLOGY
- SPORT MEDICINE
- Gold Membership
The Temporomandibular Joint
CHAPTER 26 The Temporomandibular Joint CHAPTER OBJECTIVES At the completion of this chapter, the reader will be able to: Describe the anatomy of the temporomandibular joint (TMJ), including the bones, ligaments, muscles, and blood and nerve supply. Describe the biomechanics of the TMJ, including the movements, normal and abnormal joint barriers, kinesiology, and reactions to various stresses. Summarize the various causes of temporomandibular dysfunction (TMD). Describe the close association between the TMJ, the middle ear, and the cervical spine. Perform a comprehensive examination of the temporomandibular musculoskeletal system, including palpation of the articular and soft-tissue structures, specific passive mobility, and passive articular mobility tests, and stability tests. Evaluate the total examination data to establish a diagnosis. Recognize the manifestations of abnormal TMJ function and develop strategies to correct these abnormalities. Apply active and passive mobilization techniques to the TMJ, using the correct grade, direction, and duration. Describe and demonstrate intervention strategies and techniques based on clinical findings and established goals. Evaluate the intervention effectiveness in order to progress or modify an intervention. Plan an effective home program and instruct the patient in this program. OVERVIEW Approximately 50–75% of the general population has experienced unilateral temporomandibular joint dysfunction (TMD) on a minimum of one occasion and that at least 33% have reported a minimum of one continuing persistent symptom. 1 , 2 TMD is a collective term used to describe a number of related disorders affecting the stomatognathic system and its related structures, all of which may have common symptoms. The term TMJ dysfunction as an overall descriptor of stomatognathic system dysfunction has been discontinued because it implies structural problems when none may exist, and does not include the many other factors that may be involved. 3 Housed within the skull are the components of the stomatognathic system, which includes the TMJ, the masticatory systems, and the related organs and tissues such as the inner ear and salivary glands. 4 An interrelationship exists between the stomatognathic system and the head and neck due to their proximity and shared embryological development. An understanding of this relationship is vital to understand the reasons for the myriad of symptoms that this region can exhibit. The embryologic structures from which the head, the face, and the neck originate are segmentally organized during development with the appearance and modification of six paired branchial or pharyngeal arches. 4 These branchial arches contain the cranial nuclei of the trigeminal nerve (ophthalmic; maxillary and mandibular), the facial, the glossopharyngeal, and the laryngeal branch of the vagus nerve as well as the hypoglossal nerve. The first of these arches, the mandibular arch, consists of a large anterior (ventral) part (the mandibular process of Meckel’s cartilage) and a small posterior (dorsal) (maxillary) process. As development progresses, both processes disappear except for two small portions at the posterior (dorsal) ends, which persist. The first brachial arch forms the mandible; the rudiments of the inner ear bones, the malleus, and incus; the anterior malleolar and sphenomandibular ligaments of the TMJ; the tensor tympani and the tensor veli palatini of the inner ear; the mylohyoid and the anterior belly of the digastric muscle; and the trigeminal mandibular nerve. The second pharyngeal arch (the hyoid arch) consists of Reichert’s cartilage. This arch is involved in the formation of the superior component of the hyoid bone and the lesser cornu bone; the stapes muscle; the temporal styloid process; the stylohyoid ligament; the stapedius muscle; the stylohyoid muscle; the posterior belly of the digastric muscle; the muscles of facial expression and mastication; the platysma muscle; and the glossopharyngeal nerve. The third pharyngeal arch is involved in the formation of the greater cornu of the hyoid and its body, the stylopharyngeal muscle, and the sensory apparatus of the posterior one-third of the tongue. The fourth pharyngeal arch combines with the sixth arch to form the thyroid, cricoid, and arytenoid cartilages of the larynx. The muscles derived from this arch are the pharyngeal constrictors (the cricothyroid) and the intrinsic muscles of the larynx. The pharyngeal constrictors are innervated by the superior laryngeal branch of the vagus nerve. The intrinsic muscles of the larynx are innervated by the recurrent laryngeal branch of the vagus nerve. In primitive creatures, and the human fetus, vibrations through the jaw are used as a basis for hearing. At around 8½ weeks, the small bones of the inner ear (the malleus, incus, and stapes) can be seen as distinct entities. The development of the malleus bone and the tensor tympani are intimately related to that of the lateral pterygoid muscle. Due to this embryological relationship, it is theorized that a spasm of the lateral pterygoid muscle can increase the tension within the tensor tympani (similar to that of a drum skin) 4 resulting in increased sensitivity to pitch and vibration. Theoretically, this increased tension could produce sensorineural tinnitus, or ringing in the ears, 4 a common associated symptom of a TMD, and an injury to the craniovertebral region. The diagnosis of TMD, like that of whiplash syndrome, remains controversial. 5 This is due in part to a paucity of studies regarding the incidence, course, management, and prognosis of claimed TMDs. 6 , 7 However, reports of TMD appear to be quite common. Although 50–75% of the general population has at least one sign of a TMD, only around one in four people with signs is actually aware of, or reports, any symptoms, 8 , 9 and only about 5% of people with one or more signs of a TMD will actually seek an intervention. 9–11 TMD tends to affect women more often than men, 12 specifically women over the age of 55. 13 After a technology assessment conference in 1996 about managing TMD, the National Institutes of Health (NIH) concluded that the natural history and etiology of TMD are not well understood and that most TMD symptoms are self-limiting, can recur, and may fluctuate over time. 14 The most common TMD by far, comprising 90–95% of all TMD cases, is a condition with multiple musculoskeletal facial pain complaints and a variety of jaw impairments, without an identified structural cause. 15 Due to the complex interplay between the sympathetic and trigeminal nervous systems and an increased understanding of the centralization of pain, TMD is best approached as a cluster of related disorders in the stomatognathic system that have many causes and common symptoms. 16 There are likely three etiologic factors of TMD: (1) predisposing factors, (2) precipitating or triggering factors, and (3) perpetuating or sustaining factors 17 : Predisposing factors include the structural, neurologic, hormonal, and metabolic features of an individual. Precipitating factors generally fall into the following four categories: (1) overt, extrinsic trauma to the head, the neck, or the jaw; (2) repeated low-grade extrinsic trauma, such as nail biting and chewing gum; (3) repeated low-grade intrinsic trauma such as teeth clenching or bruxism (grinding teeth); and (4) stress that passes a certain threshold, which is individual for each patient. Perpetuating or contributing factors are those that aid in the continuation of symptoms. These can include systemic disease and cervical pathology. Thus, the clinical course of TMD does not reflect a progressive disease but rather a complex disorder that is molded by many interacting factors, such as stress, anxiety, and depression, which serve to maintain the disease. 16 Headaches, orofacial pain, earache, and neck pain are common complaints. A diagnosis of TMD must, therefore, include an examination of all of the following: Jaw muscles Bone and cartilage joint structures Facial structures Soft-tissue joint structures, including the articular disk and synovium Jaw and joint function Cervical and upper thoracic spine function Posture and dysfunction Systemic disease Psychosocial issues Given the number of potential causes of jaw and face pain, a diagnosis of TMD can rarely be ascribed solely to the TMJ. Examples of appropriate diagnoses for TMD are more likely to include rheumatoid arthritis with synovitis, arthralgia, condylar degenerative disease, and open bite deformity; chronic pain with a behavioral disorder; myofascial pain and impairment; and internal disk derangement, with displacement and reduction. CLINICAL PEARL Most of those who seek medical intervention for TMDs are female. The reason for the higher prevalence of TMD in women, and the overrepresentation of females at orofacial pain clinics remains obscure. One explanation could be that women more readily seek treatment for illness than do men. 18
Nonsurgical interventions such as counseling, physical therapy, pharmacotherapy, and occlusal splint therapy continue to be the most effective way of managing more than 80% of patients with TMD. 16 Although dentists are the primary professionals involved in the examination and intervention of TMD, physical therapists can play an important role in assisting the dentist in restoring function to the stomatognathic system. However, the procedures in physical therapy intervention are not well described in the literature in this area. ANATOMY The TMJ ( Fig. 26-1 ) is a synovial, compound, modified ovoid bicondylar joint, which is formed between the articular eminence of the temporal bone, the intra-articular disk, and the head of the mandible. FIGURE 26-1 The temporomandibular joint. (Reproduced, with permission, from Chapter 21 . Infratemporal Fossa. In: Morton DA, Foreman K, Albertine KH. eds. The Big Picture: Gross Anatomy. New York, NY: McGraw-Hill; 2011.) The TMJ is unique in that, even though the joint is synovial, the articulating surfaces of the bones are covered not by hyaline cartilage but by fibrocartilage. 19 , 20 Fibrocartilage has the same general properties found in hyaline cartilage but tends to be less distensible, owing to a greater proportion of dense collagen fibers (see Chapter 1 ). The development of fibrocartilage over the load-bearing surface of the TMJ indicates that the joint is designed to withstand large and repeated stresses and that this area of the joint surface has a greater capacity to repair itself than would hyaline cartilage. 21 The area of load bearing is affected by the congruity of the contacting tooth surfaces (occlusion), head position, and the coordination of muscle function. The fibrocartilage is at its thinnest at the roof of the fossa, but load bearing here occurs only in the presence of dysfunction. 22 The mandible works like a class-three lever (see Chapter 1 ), with its joint as the fulcrum. Although there is no agreement among the experts concerning force transmission through the joint, there does appear to be agreement that postural impairments of the cervical and upper thoracic spine can produce both pain and impairment of the TMJ. 8 Bony Anatomy
A number of bony components make up the masticatory system: the maxilla and the mandible, which support the teeth, and the temporal bone, which supports the mandible at its articulation with the skull. The sphenoid bone and the hyoid bone also could be included, because they provide important anatomic and functional links to the TMJ. Maxilla
The borders of the maxillae extend superiorly to form the floor of the nasal cavity as well as the floor of each orbit ( Fig. 26-1 ). Inferiorly, the maxillary bones form the palate and the alveolar ridges, which support the teeth. The maxillae and mandible each contains 16 permanent teeth. The structure of each tooth reflects its function in mastication. Sphenoid Bone
The greater wings of the sphenoid bone form the boundaries of the anterior part of the middle cranial fossa. From these greater wings, the pterygoid laminae serve as the attachments for the medial and lateral pterygoid muscles. Hyoid Bone
The U-shaped hyoid bone ( Fig. 26-2 ), also known as the skeleton of the tongue, serves as the attachment for the infrahyoid muscles and for some of the extrinsic tongue muscles. The hyoid bone is involved with the mandible to provide reciprocal stabilization during swallowing and chewing. Theoretically, due to its muscle attachments, the position of the hyoid bone can be affected by cervical and shoulder positions, as occurs during forward head posture, which changes the length–tension relationships. FIGURE 26-2 The hyoid bone and related muscles. (Reproduced, with permission, from Chapter 25 . Overview of the Neck. In: Morton DA, Foreman K, Albertine KH. eds. The Big Picture: Gross Anatomy . New York, NY: McGraw-Hill; 2011.) Mandible
The mandible, or jaw ( Fig. 26-1 ), which supports the lower teeth, is the largest and the strongest bone in the face. It is suspended below the maxillae by muscles and ligaments that provide it with both mobility and stability. The medial surface of the mandible serves as the attachment for the medial pterygoid and the digastric muscles. The platysma, mentalis, and buccinator gain attachment on its lateral aspect. Two broad, vertical rami extend upward from the mandible: the condyle and the coronoid process. The anterior of the two processes, the coronoid, serves as the attachment for the temporalis and masseter muscles. 23 The condyle process articulates with the temporal bone. The bony surfaces of the condyle and the articular portion of the temporal bone are made of dense cortical bone. The articulating surface of the condyle is flattened from front to back with its medial–lateral length twice as long as its anterior–posterior length. The condyles are generally convex, possessing short bony projections known as medial and lateral poles. 24 Temporal Bone
The mandibular fossa of the temporal bone is divided into two surfaces: articular and nonarticular. 24 The articulating surface of the temporal bone is made up of a concave mandibular, or glenoid, fossa and a convex bony prominence called the articular eminence. 25 The articular tubercle, situated anterior to the glenoid fossa, serves as an attachment for the temporomandibular (or lateral) ligament. 23 The nonarticular surface of the fossa consists of a very thin layer of bone and fibrocartilage that occupies much of the superior and posterior walls of the fossa. 24 Fibrocartilaginous Disk
Located between the articulating surface of the temporal bone and the mandibular condyle is a fibrocartilaginous disk (sometimes inappropriately referred to as “meniscus”) ( Fig. 26-3 ). The biconcave shape of the disk is determined by the shape of the condyle and the articulating fossa. 26 Rees 20 has described the fibrocartilaginous disk as having three clearly defined transverse, ellipsoidal zones that are divided into three regions—posterior band, intermediate zone, and anterior band—of which the intermediate zone makes contact with the articular surface of the condyle. FIGURE 26-3 The TMJ disk and the pterygoid. (Reproduced, with permission, from Chapter 21 . Infratemporal Fossa. In: Morton DA, Foreman K, Albertine KH. eds. The Big Picture: Gross Anatomy . New York, NY: McGraw-Hill; 2011.) Both the disk and the lateral pterygoid muscle develop from the first branchial arch, and there is very little differentiation among the muscle, the disk, and the joint capsule. 27 , 28 The fibrocartilaginous disk is tethered by a number of structures: Medial and lateral collateral diskal ligaments firmly attach the fibrocartilaginous disk to the medial and lateral poles of the condyle, permitting anterior and posterior rotation of the disk on the condyle during mouth opening and closing. 29 , 30 Posteriorly, the disk is attached by fibroelastic tissue to the posterior mandibular fossa and the back of the mandibular condyle. 29 , 30 Anteriorly, the disk is attached to the upper part of the tendon of the lateral pterygoid muscle ( Fig. 26-3 ). 29 , 30 The disk usually is located on top of the condyle in the 12 o’clock to 1 o’clock position on the mandibular head when the jaw is closed. 31 Since the only firm attachment of the disk to the condyle occurs medially and laterally, the disk can move somewhat independently of the condyle. 32 The disk effectively divides the TMJ into a lower and an upper joint cavity (see Fig. 26-3 ): Lower compartment. This compartment, bordered by the mandibular condyle and the inferior surface of the articular disk, is where, under normal conditions, the osteokinematic spin (rotation) of the condyle occurs. 8 Upper compartment. This compartment, bordered by the mandibular fossa and the superior surface of the articular disk, primarily allows only translation of the disk and condyle along the fossa, and onto the articular eminence. 8 Blood vessels and nerves are found only in the thickened periphery of this disk, especially its posterior attachment; its middle articular portion is avascular and aneural. 33 Supporting Structures
The supporting structures of the TMJ consist of periarticular connective tissue (ligament, tendon, capsule, and fascia). As its name implies, the periarticular connective tissue serves to keep the joints together and to limit the ranges of motion at the joint. For example, the ligaments of the TMJ protect and support the joint structures and act as passive restraints to joint movement. The synovial cavities are surrounded by loose connective tissue rather than by ligaments. The intercapsular structures are located posteriorly to the condyle. Anterior to the joint are the muscles of the medial and lateral pterygoid (see next section). There are no well-defined anterior or posterior ligaments between the mandibular condyle and the temporal bone. However, two strong ligaments help to provide joint stability: Joint capsule or capsular ligament. This structure, which surrounds the entire joint, is thought to provide proprioceptive feedback regarding the joint position and movement. 34 , 35 Temporomandibular (or lateral) ligament. The capsule of the TMJ is reinforced laterally by an outer oblique portion and an inner horizontal portion of the temporomandibular ligament, which function as a suspensory mechanism for the mandible during moderate opening movements. The ligament also functions to resist rotation and posterior displacement of the mandible. Two other ligaments assist with joint stability: Stylomandibular ligament. The stylomandibular ligament is a specialized band that splits away from the superficial lamina of the deep cervical fascia to run deep to both pterygoid muscles. 25 This ligament becomes taut and acts as a guiding mechanism for the mandible, keeping the condyle, disk, and temporal bone firmly opposed. Sphenomandibular ligament. The sphenomandibular ligament is a thin band that runs from the spine of the sphenoid bone to a small bony prominence on the medial surface of the ramus of the mandible, called the lingula. This ligament acts to check the angle of the mandible from sliding as far forward as the condyles during the translatory cycle and serves as a suspensory ligament of the mandible during wide opening. 25 It is this ligament that hurts with any prolonged jaw opening, such as that which occurs at the dentist. CLINICAL PEARL Pinto’s ligament 36 is a vestige of Meckel’s cartilage, an embryological tissue. It arises from the neck of the malleus of the inner ear and runs in a medial-superior direction to insert into the posterior aspect of the TMJ capsule and disk. While the role of this ligament in mandibular mechanics is thought to be negligible, its relationship to the middle ear and the TMJ could be a basis for the middle ear symptoms, which are often present with TMD. Stack and Funt 37 postulated that auditory symptoms of fullness and pressure in the middle ear might be present as a result of direct transmission of TMJ capsular tension to the ossicles of the middle ear through this ligament. Muscles
The muscles of mastication are the key muscles when discussing TMD. Three of these muscles, the masseter, the medial pterygoid, and the temporalis ( Fig. 26-4 ), function to raise the mandible during mouth closing. The lateral pterygoid and digastric muscles work together to depress the mandible during mouth opening. FIGURE 26-4 Lateral view of TMJ. (Reproduced, with permission, from Chapter 21 . Infratemporal Fossa. In: Morton DA, Foreman K, Albertine KH. eds. The Big Picture: Gross Anatomy. New York, NY: McGraw-Hill; 2011.) Although these muscles work most efficiently in groups, an understanding of the specific anatomy and action(s) of the individual muscles is necessary for an appreciation of their coordinated function during masticatory activity ( Tables 26-1 and 26-2 ). TABLE 26-1 Muscles of the Temporomandibular Joint Muscle Proximal Distal Innervation Medial pterygoid Medial surface of lateral pterygoid plate and tuberosity of maxilla Medial surface of mandible close to angle Mandibular division of trigeminal nerve Lateral pterygoid Greater wing of sphenoid and lateral pterygoid plate Neck of mandible and articular cartilage Mandibular division of trigeminal nerve Temporalis Temporal cranial fossa By way of a tendon into medial surface, apex, and anterior and posterior borders of mandibular ramus Anterior and posterior deep temporal nerves, which branch from anterior division of mandibular branch of trigeminal nerve Masseter Superficial portion: from anterior two-thirds of lower border of zygomatic arch; deep portion from medial surface of zygomatic arch Lateral surfaces of coronoid process of mandible, upper half of ramus, and angle of mandible Masseteric nerve from anterior trunk of mandibular division of trigeminal nerve Mylohyoid Medial surface of mandible Body of hyoid bone Mylohyoid branch of trigeminal nerve and mandibular division Geniohyoid Mental spine of mandible Body of hyoid bone Anterior (ventral) ramus of C1 via hypoglossal nerve Stylohyoid Styloid process of temporal bone Body of hyoid bone Facial nerve Anterior and posterior digastric Internal surface of mandible and mastoid process of temporal bone By intermediate tendon to hyoid bone Anterior: mandibular division of trigeminal nerve; posterior: facial nerve Sternohyoid Manubrium and medial end of clavicle Body of hyoid bone Ansa cervicalis Omohyoid Superior angle of scapula Inferior body of hyoid bone Ansa cervicalis Sternothyroid Posterior surface of manubrium Thyroid cartilage Ansa cervicalis Thyrohyoid Thyroid cartilage Inferior body and greater horn of hyoid bone C1 via hypoglossal nerve TABLE 26-2 Actions of the Temporomandibular Joint Muscles Action Muscles Acting Opening of mouth Lateral pterygoid Mylohyoid Geniohyoid Digastric Closing of mouth Masseter Temporalis Medial pterygoid Protrusion of mandible Lateral pterygoid Medial pterygoid Masseter Mylohyoid Geniohyoid Digastric Stylohyoid Temporalis (anterior fibers) Retraction of mandible Temporalis (posterior fibers) Masseter Digastric Stylohyoid Mylohyoid Geniohyoid Lateral deviation of mandible Lateral pterygoid (ipsilateral muscle) Medial pterygoid (contralateral muscle) Temporalis Masseter Temporalis
The temporalis muscle ( Fig. 26-4 ) has its origin from the floor of the temporal fossa and the temporal fascia. The muscle travels inferiorly and anteriorly to insert on the anterior border of the coronoid process and the anterior border of the ramus of the mandible. The temporalis muscle is innervated by a branch of the mandibular division of the trigeminal nerve. In addition to assisting with mouth closing and side-to-side grinding of the teeth, the temporalis muscle provides a good deal of stability to the joint. The Masseter
The masseter ( Fig. 26-4 ) is a two-layered quadrilateral-shaped muscle. The superficial portion arises from the anterior two-thirds of the lower border of the zygomatic arch. The deep portion arises from the medial surface of the zygomatic arch. Both sets of fibers blend anteriorly and form a raphe with the medial pterygoid. 25 The masseter inserts on the lateral surface of the coronoid process of the mandible, the upper half of the ramus and angle of the mandible. The masseter muscle is innervated by a branch of the mandibular division of the trigeminal nerve. The major function of the masseter is to elevate the mandible, thereby occluding the teeth during mastication. The Medial Pterygoid
The medial pterygoid muscle is a thick quadrilateral muscle, with a deep origin situated on the medial aspect of the mandibular ramus ( Fig. 26-4 ). The muscle travels posteriorly to insert on the inferior and posterior aspects of the medial subsurface of the ramus and angle of the mandible. The medial pterygoid muscle is innervated by a branch of the mandibular division of the trigeminal nerve. Working bilaterally, and in association with the masseter and temporalis muscles, the medial pterygoids assist in mouth closing. Individually, the medial pterygoid muscle is capable of deviating the mandible toward the opposite side. The medial pterygoid muscle also assists the lateral pterygoid and anterior fibers of the temporalis muscle to produce protrusion of the mandible. The Lateral Pterygoid
Two divisions of the lateral pterygoid muscles are recognized, each of which is functionally and anatomically separate ( Figs. 26-3 and 26-4 ). The superior head arises from the infratemporal surface of the greater wing of the sphenoid. The inferior head arises from the lateral surface of the lateral pterygoid plate. Despite several investigations, 38–40 no consensus has been reached regarding the insertion of the lateral pterygoid muscle. However, the most commonly described insertion is at the anterior aspect of the neck of the mandibular condyle and capsule of the TMJ. The lateral pterygoid muscle is innervated by a branch of the mandibular division of the trigeminal nerve. The superior head of the lateral pterygoid is involved mainly with chewing and functions to rotate the disk anteriorly on the condyle during the closing movement. 41 , 42 It has also been suggested that in normal function of the craniomandibular complex, the superior lateral pterygoid plays an important role in stabilizing and controlling the movements of the disk. 43 The inferior head of the lateral pterygoid muscle exerts an anterior, lateral, and inferior pull on the mandible, thereby opening the jaw, protruding the mandible, and deviating the mandible to the opposite side. Infrahyoid or “Strap” Muscles
The infrahyoid muscles comprise the sternohyoid, omohyoid, sternothyroid, and thyrohyoid muscles ( Fig. 26-5 ). FIGURE 26-5 The infrahyoid muscles. (Reproduced, with permission, from Chapter 25 . Overview of the Neck. In: Morton DA, Foreman K, Albertine KH. eds. The Big Picture: Gross Anatomy . New York, NY: McGraw-Hill; 2011.) Sternohyoid The sternohyoid muscle is a strap-like muscle that functions to depress the hyoid and assist in speech and mastication. Omohyoid The omohyoid muscle, situated lateral to the sternohyoid, consists of two bellies and functions to depress the hyoid. In addition, the muscle has been speculated to tense the inferior aspect of the deep cervical fascia in prolonged inspiratory efforts, thereby releasing tension on the apices of the lungs and on the internal jugular vein, which are attached to this fascial layer. 25 Sternothyroid and Thyrohyoid The sternothyroid and thyrohyoid muscles (see Fig. 26-5 ) are located deep to the sternohyoid muscle. The sternothyroid muscle is involved in drawing the larynx downward, whereas the thyrohyoid depresses the hyoid and elevates the larynx. These infrahyoid muscles are innervated by fibers from the upper cervical nerves. The nerves to the lower part of these muscles are given off from a loop, the ansa cervicalis (cervical loop) (see Chapter 3 ). Suprahyoid Muscles
The suprahyoid muscles ( Fig. 26-2 ), working with the infrahyoid muscles, play a major role in coordinating mandibular function, by providing a firm base on which the tongue and mandible can be moved. Geniohyoid The geniohyoid muscle is a narrow muscle situated under the mylohyoid muscle. The muscle functions to elevate the hyoid bone. Digastric As its name suggests, the digastric muscle consists of two bellies. The posterior belly arises from the mastoid notch of the temporal bone while the anterior belly arises from the digastric fossa of the mandible. The posterior belly is innervated by a branch of the facial nerve. The anterior belly is innervated by the inferior alveolar branch of the trigeminal nerve. The two bellies of the digastric muscle are joined by a rounded tendon that attaches to the body and greater cornu of the hyoid bone through a fibrous loop or sling. 25 Bilaterally, the two bellies of the digastric muscle assist in forced mouth opening by stabilizing the hyoid. The posterior bellies are especially active during coughing and swallowing. 25 CLINICAL PEARL The muscles of the TMJ, working in combinations, are involved as follows: Mouth opening—bilateral action of the lateral pterygoid and digastric muscles. Mouth closing—bilateral action of the temporalis, masseter, and medial pterygoid muscles. Lateral deviation—action of the ipsilateral masseter, and contralateral medial and lateral pterygoid muscles. Protrusion—bilateral action of the lateral pterygoid, medial pterygoid, and anterior fibers of the temporalis muscles. Retrusion—bilateral action of the posterior fibers of the temporalis muscle, the digastric, stylohyoid, geniohyoid, and mylohyoid muscles. Mylohyoid This flat, triangular muscle is functionally a muscle of the tongue, stabilizing or elevating the tongue while swallowing and elevating the floor of the mouth in the first stage of deglutition. 25 Stylohyoid The stylohyoid muscle elevates the hyoid and base of the tongue and has an undetermined role in speech, mastication, and swallowing. Nerve Supply
The TMJ is primarily supplied by three nerves that are part of the mandibular division of the fifth cranial (trigeminal) nerve ( Fig. 26-6 ) ( Box 26-1 ). Portions of the middle ear ossicles, middle ear musculature, and muscles of mastication all originate from the first branchial arch and are innervated by this nerve. Therefore, in a patient with altered bite mechanics, spasm of the muscles of mastication caused by a displaced condyle may cause neuromuscular dysfunction of all the muscles innervated by the trigeminal nerve, including the tensor palatini. 44 FIGURE 26-6 The trigeminal nerve. (Reproduced, with permission, from Chapter 17 . Cranial Nerves. In: Morton DA, Foreman K, Albertine KH. eds. The Big Picture: Gross Anatomy . New York, NY: McGraw-Hill; 2011.) Box 26-1 Characteristics of the Trigeminal Nerve Motor Nucleus The anterolateral upper pons. Sensory Nucleus There are two nuclei: (1) the chief sensory nucleus in the posterior (dorsal)-lateral pons and (2) the mesencephalic nucleus, which extends from the chief sensory nucleus upward through the pons to the midbrain. Spinal Nucleus The spinal tract consists of small- and medium-sized myelinated nerve fibers and runs caudally to reach the upper cervical segments of the spinal cord. The lowest nerve fibers in the tract mix with the spinal fibers in the tract of Lissauer. Nerves Mandibular Ophthalmic Maxillary Termination Muscles of mastication, both pterygoids, tensor veli palatini, tensor tympani, mylohyoid, and anterior belly of digastric. Skin of vertex, temporal area, forehead, and face; mucosa of sinuses, nose, pharynx, anterior two-thirds of tongue, and oral cavity. Lacrimal, parotid, and lingual glands; dura of anterior and middle cranial fossae. External aspect of tympanic membrane and external auditory meatus, TMJ, and teeth. Dilator pupillae and probably proprioceptors of extraocular muscles. Sensation from upper three or four cervical levels.
There is considerable clinical interest in the interactions between the cervical and craniofacial regions. This interest stems from a number of reports concerning patients who have pain in the cervical and craniofacial areas simultaneously. 32 , 45–48 In the suboccipital region, a series of dense neural connections, called the trigeminocervical complex, exists among trigeminal, facial, glossopharyngeal, and vagus nerves, with those of the upper C1–4 cervical spinal nerves ( Fig. 26-7 ). 17 Postural abnormalities resulting from various acute or chronic etiologies that produce suboccipital compression may, therefore, be responsible for craniofacial pain anywhere in the head, in addition to symptoms of dizziness or nystagmus. 7 , 17 , 49 FIGURE 26-7 Neurology of the TMJ. (Reproduced, with permission, from Chapter 21 . Infratemporal Fossa. In: Morton DA, Foreman K, Albertine KH. eds. The Big Picture: Gross Anatomy . New York, NY: McGraw-Hill; 2011.) TMJ-related headaches usually include pain near the TMJ and ear, ear fullness, temporal headaches, and facial pain. 50 The dizziness associated with TMD tends to be of the nonvertiginous variety, with the patient complaining of unsteadiness, giddiness, or lightheadedness. 51 Although the exact mechanism is unclear, postural influences, alteration in the position of the jaw by the malocclusion, and the subsequent mismatching between the cervical muscles might be the cause. 51 BIOMECHANICS The movements that occur at the TMJ are extremely complex. The TMJ has three degrees of freedom, with each of the degrees of freedom being associated with a separate axis of rotation. 52 Two primary arthrokinematic movements (rotation and anterior translation) occur at this joint around three planes: sagittal, horizontal, and frontal. Rotation occurs from the beginning to the midrange of movement. In addition to the rotational motions during mouth opening and closing and lateral deviations, movements at the TMJ involve arthrokinematic rolls and slides. Gliding, translation, or sliding movements occur in the upper cavity, whereas rotation or hinged movement occurs in the lower cavity. The motions of protrusion and retrusion are planar glides. Thus, mouth opening, contralateral deviation, and protrusion all involve an anterior osteokinematic rotation of the mandible and an anterior, inferior, and lateral glide of the mandibular head and disk; mouth closing, ipsilateral deviation, and retrusion all involve a posterior osteokinematic rotation of the mandible and an anterior, inferior, and lateral glide of the mandibular head and disk. Occlusal Position
Occlusal positions are functional positions of the TMJ. The occlusal position is defined as the point at which contact between some or all of the teeth occurs. Under normal circumstances, the upper molars rest directly on the lower molars and the upper incisors slightly override the lower incisors. The ideal position provides mutual protection of the anterior and posterior teeth, comfortable and painless mandibular function, and stability. 17 The median occlusal position corresponds to the position in which all of the teeth are fully interdigitated 19 and is considered the start position for all mandibular motions. The median occlusal position is dependent on the presence, shape, and position of the teeth. Protrusion of the upper or lower incisors, failure of the upper incisors to overlap with the lower incisors, absent or abnormally shaped teeth, and back teeth that do not meet are all causes of malocclusion. The centric position is considered to be the position that implies the most retruded, unstrained position of the mandible from which lateral movements are possible, and the components of the oral apparatus are the most balanced. 53 Ideally, the centric position should coincide with the median occlusal position. 53 It is worth remembering that malocclusion is probably very common in the general nonsymptomatic patient and may or may not be relevant to the presenting symptoms. 54 Rather than being a primary etiologic factor in TMD, malocclusion is likely to have a secondary or contributory role. 17 , 55 Mouth Opening
Mouth opening occurs in a series of steps ( Table 26-3 ). In the erect position, the condyles begin to rotate anteriorly and translate inferiorly and laterally during the first 25 degrees of opening as the jaw opens. The upper head of the lateral pterygoid muscle and the anterior head of the digastric muscle draw the disk anteriorly and prepare for condylar rotation during movement. 32 This initial condylar rotation occurs as the mandibular elevators (masseter, temporalis, and medial pterygoid muscles) gradually relax and lengthen, allowing gravity to depress the mandible (see Fig. 26-8 ). 32 The directions of the fibers of the lateral and medial temporomandibular ligaments also keep the condyle from moving posteriorly. The fibrous capsule and parts of the temporomandibular ligament limit excessive lateral movement of the condyle. The rotation occurs through the two condylar heads between the articular disk and the condyle. As the mandible moves forward on opening, the disks move medially and posteriorly until the collateral ligaments, and the lateral pterygoid stop their movement. During the last 15 degrees of opening, the rotation ceases due to tightening of the collateral ligaments, and is replaced by an anterior translation of the condyles (see Fig. 26-8 ). 56 During this translation, the condyle and disk move together. The anterior translation, which is produced mainly by muscle contraction, serves to prevent mandibular encroachment of the anterior neck structures. Opening is also assisted by the other suprahyoid muscles. 32 In extremely wide opening, such as that occurs with yawning, the functional joint contact is on the distal aspect of the condyle, and the anterior lateral aspect of the condyle contacts the posterior part of the masseter muscle. In this position, the soft-tissue structures are in a position of stretch, making them more prone to dysfunction. 57 FIGURE 26-8 Mouth opening and closing. TABLE 26-3 Arthrokinematic Steps of the Temporomandibular Joint Step Movement Rest position Joint is in an open-packed position Rotation There is a mid-opening Condylar joint surfaces glide forward, inferior joint surface of disk has a relative posterior glide, upper lateral pterygoid relaxes, inferior pterygoid contracts, and posterior connective tissue is in a functional state of rest Functional opening Disk and condyle experience a short anterior translatory glide; superior and inferior heads of lateral pterygoid contract to guide disk and condyle forward Posterior connective tissue is in a functional tightening Translation There is full opening Disk and condyle glide anteriorly and caudally Superior and inferior heads of lateral pterygoid contract to guide disk and condyle fully forward Posterior connective tissues tighten Closure Surface of condyle joint glides posteriorly, and disk glides relative to anterior surface Superior head of lateral pterygoid contracts and inferior head relaxes Posterior connective tissue returns to its functional length Data from Rocabado M. Arthrokinematics of the temporomandibular joint. In: Gelb H, ed. Clinical Management of Head, Neck and TMJ Pain and Dysfunction. Philadelphia, PA: WB Saunders; 1985. Mouth Closing
Closing of the mouth involves a reversal of the movements described for mouth opening. The condyles translate posteriorly as a result of an interaction between the retracting portions of the masseter and temporalis muscle and the retracting portions of the mandibular depressors (see Fig. 26-8 ). 52 As the condyles translate posteriorly and glide medially, they hinge on the disks. The disks then glide posteriorly and superiorly on the temporal bone along with the condyles (as a result of the actions of the masseter, medial pterygoid, and temporalis muscles). 42 When the jaws are closed to maximal occlusal contact, the condyles contact the disks, and the disks contact the posterior slopes of the articular tubercles and the glenoid fossae. Protrusion
Protrusion is a forward movement of the mandible that occurs at the superior joint compartments, which consists of the disk and condyle moving downward, forward, and laterally. The muscles responsible for protrusion are the anterior fibers of the temporalis and the medial and lateral pterygoid muscles. Retrusion
Retrusion is a backward movement of the mandible, produced by the posterior fibers of the temporalis and assisted by the suprahyoid muscles. The retrusive range is limited by the extensibility of the temporomandibular ligaments. 58 Lateral Excursion
If a protrusion movement occurs unilaterally, it is called a lateral excursion, or deviation. For example, if only the left TMJ protrudes, the jaw deviates to the right. Lateral movements of the mandible are the result of asymmetric muscle contractions ( Fig. 26-9 ). During a lateral excursion to the right, the condyle and the disk on the left side glide inferiorly, anteriorly, and laterally in the sagittal plane and medially in the horizontal plane along the articular eminence. The condyle and the disk on the right side rotate laterally on a sagittal plane and translate medially in the horizontal plane while remaining in the fossa. FIGURE 26-9 Lateral deviation to the left. CLINICAL PEARL The translation of the human condyle during jaw opening and lateral jaw movements is referred to as the Bennett shift. The Close- and Open-Packed (Resting) Positions
The close-packed position of the TMJ is difficult to determine because the position of maximal muscle tightness is also the position of least joint surface congruity and vice versa. Rocabado 56 considers there to be two close-packed positions, named according to the end position of the mandibular head in the fossa: Anterior close-packed position. This position is the position of maximum opening of the joint. Posterior close-packed position. This position is the maximum retruded position of the joint. Under this premise, the open-packed, or “rest” position is any position away from the anterior or posterior close-packed positions of the joint. 56 The rest position, or “freeway space” corresponds to the position of the TMJ where the residual tension of the muscles is at rest and no contact occurs between maxillary and mandibular teeth. In this position, the tongue is against the palate of the mouth with its most anterior–superior tip in the area just posterior to the rear of the upper central incisors. 59 Capsular Pattern
The capsular pattern of the TMJ is a limitation of mouth opening. If one joint is more involved than the other, the jaw will laterally deviate to the same side during opening. CLINICAL PEARL The significance of the rest position is that it permits the tissues of the stomatognathic system to rest and undergo repair. 60 EXAMINATION Currently, clinical examination is the gold standard for diagnosing TMDs. Given the multifactorial causes of TMD, a comprehensive examination of the entire upper quadrant, including the cervical spine and shoulders, usually is warranted. In general, the TMJ and the upper three cervical joints all refer symptoms to the head, whereas the mid-to-low cervical spine typically refers symptoms to the shoulder and the arm. 61–63 An accurate diagnosis of TMD involves a careful evaluation of the information gleaned from the history, systems review, and tests and measures. In most chronic cases, a behavioral or psychological examination is required. 3 , 31 , 63–68 Since postural dysfunctions are closely related to TMJ symptoms, the clinician should always perform a postural examination as part of a comprehensive examination of this joint. An examination form for the TMJ examination is shown in Table 26-4 . TABLE 26-4 Temporomandibular Examination Form History
A comprehensive history will help identify the possible source(s) of the orofacial pain and provide a screen for other causative or contributing factors. 69 During the history, the clinician should observe whether the patient’s mouth moves comfortably. The severity of the symptoms and the time before the symptoms subside can provide the clinician with valuable information regarding possible pathology. Red flags related to cardiac history (e.g., angina or history of myocardial infarction) and brain function (e.g., sudden-onset severe headaches, weakness, or slurred speech) must be investigated early in the history taking. 70 The clinician should determine from the patient the main reason for the visit. There are three cardinal features of TMDs, which can be local or remote: Restricted jaw function. Limited mouth opening, which may be reported as intermittent or progressive, is a key feature of TMD. A key question to determine restricted jaw function is, “Have you ever had your jaw lock or catch so that it would not open all the way? If so, was this limitation in jaw opening severe enough to interfere with your ability to eat? Have you ever noticed clicking, or popping or other sounds in your joint?” 71 Patients may describe a generalized tight feeling, which may indicate a muscular disorder, or capsulitis, or the sensation that the jaw suddenly “catches” or “locks,” which usually is related to a mechanical interference within the joint (an internal derangement). 16 Associated signs of an internal derangement include pain and deviation of mandibular movements during opening and closing (refer to Practice Pattern 4D, under “Intervention Strategies” section later), and biting firm objects. Pain in the fully open position is probably caused by an extra-articular problem. Locking may imply that the mouth does not fully open or does not fully close and is often related to problems of the disk or joint degeneration. A gradual onset of symptoms after minor or prolonged physical activity may be indicative of a mechanical derangement. Symptoms of a mechanical nature generally are eased with rest. The irritability of a disorder is determined by the degree of activity necessary to provoke a symptom response. Joint noises. Joint noises (crepitus) of the TMJ may or may not be significant, because joint sounds occur in approximately 50% of healthy populations. 72 Some joint sounds are not audible to the clinician, so a stethoscope may be required. “Hard” crepitus is a diffuse sustained noise that occurs during a significant portion of the opening or closing cycle, or both and is an evidence of a change in osseous contour. 57 Clicking describes a brief noise that occurs at some point during opening, closing, or both [see the discussion of range of motion (ROM) testing, later]. Jaw clicking during mouth opening or closing may be suggestive of an internal derangement consisting of an anterior disk displacement with reduction. 73 , 74 Orofacial pain. TMJ pain should be evaluated carefully in terms of its onset, nature, intensity, site, duration, aggravating and relieving factors, and how it relates to joint noise and restricted mandibular movements. Information about the nature of the pain will be critical in determining the possibility of primary headaches (migraine, cluster) and secondary headaches related to the eyes, ears, sinus, dental structures, medication complications, and/or neurologic types of pain. Unrelenting pain unrelated to musculoskeltal function is an indication for referral. Key questions have been examined and determined to have strong sensitivity and specificity in incriminating TMDs as the source of pain. 76 The initiating question should be, “Have you had pain or stiffness in the face, jaw, temple, in front of the ear, or in the ear in the past month?” 70 A positive response should be followed by a question about whether the symptoms are altered by any of the following jaw activities: chewing, talking, singing, yawning, kissing, moving the jaw. 69 Specific questions about activities and postures of a sustained nature, such as sitting, sleeping, and driving, should be asked. Orofacial pain associated with mouth opening or closing and jaw crepitus is suggestive of osteoarthrosis, capsulitis, or internal derangement consisting of an anterior disk displacement with reduction. 73 , 74 Information about cervical dysfunction is essential to determine whether the cervical spine is causing or exacerbating the headache/facial pain. 70 In a study by Magnusson et al., 77 five different scales of self-assessment of pain were tested in patients with TMJ disorders. The precision and sensitivity and the capacity to register memory of pain and discomfort were compared for each of the five scales ( Table 26-5 ). From these results, the behavior rating scale can be recommended when measuring pain and discomfort in patients with TMJ disorders. TABLE 26-5 Patient Reports of Pain in TMJ Disorders Test and Measure Test Procedure Population Reliability Kappa Values Visual analog scale 100-mm line with the end defined as no pain and worst pain imaginable 38 consecutive patients referred with TMJ disorders κ = 0.38 Numeric scale 10-point scale with 0 indicating no paint and 10 representing worst pain κ = 0.36 Behavior rating scale Six-point scale ranging from minor discomfort to very strong discomfort κ = 0.68 Verbal scale Five-point scale ranging from no pain to very severe pain κ = 0.44 Data from Magnusson T, List T, Helkimo M. Self-assessment of pain and discomfort in patients with temporomandibular disorders: a comparison of five different scales with respect to their precision and sensitivity as well as their capacity to register memory of pain and discomfort. J Oral Rehabil. 1995;22:549–556.; Cleland J. Temporomandibular Joint, Orthopaedic Clinical Examination: An Evidence-Based Approach for Physical Therapists . Carlstadt, NJ. Icon Learning Systems, LLC; 2005:39–89. With permission from Icon Learning Systems. In addition, it is important to determine: If the presenting symptoms were caused by trauma or surgery, or if the onset of pain occurred gradually. Questions should focus on any history of trauma during birth or childhood, as well as more recently. If there are any emotional factors in the patient’s background that may provoke habitual protrusion or muscular tension. Chronic head, neck, and back pain often are associated with psychogenic causes. Psychiatric disorders, usually, are manifested in patients whose afflictions seem to be excessive or persist beyond what would be normal for that condition. The clinician should listen for reports of psychological stress overload, malaise, anxiety, sleep problems, changes in eating patents, weight changes, unexplained fatigue, and other signs of depression, which might exacerbate pain through central mechanisms. 79 The checklist outlined in Table 26-6 can be used by the clinician to identify factors that may warrant an examination by a mental health professional. TABLE 26-6 Checklist of Psychological and Behavioral Factors Inconsistent, inappropriate, or vague reports of pain Overdramatization of symptoms Symptoms that vary with life events Significant pain of >6 months’ duration Repeated failures with conventional therapies Inconsistent response to medications History of other stress-related disorders Major life events (e.g., new job, marriage, divorce, and death) Evidence of drug abuse Clinically significant anxiety or depression Evidence of secondary gain Note: The significance of these factors depends on the particular patient. Data from McNeill C, Mohl ND, Rugh JD, et al. Temporomandibular disorders: Diagnosis, management, education, and research. J Am Dent Assoc. 1990;120:253–260. If the patient is aware of any parafunctional habits (cheek biting, nail biting, pencil chewing, teeth clenching, or bruxism). For example, does the patient chew on one side more than the other? Chewing more on one side versus the other is typically the result of malocclusion (see later). In addition, favoring one side can lead to a loss of vertical dimension (the distance between any two arbitrary points on the face). A simple way to measure the aforementioned is to measure from the lateral edge of the eye to the corner of the mouth and from the base of the nose to the chin. The behavior of symptoms over a 24-hour period. This information assists the clinician in formulating causal relationships. Whether the symptoms are improving or worsening. The relationship of eating to the symptoms. Alcohol, chocolate, and other foods such as ice cream can cause head pain in some individuals, suggesting a vasomotor-related pain. The patient’s past dental and orthodontic history. CLINICAL PEARL TMJ sounds should be described and related to symptoms. Joint noise is, of itself, of little clinical importance in the absence of pain. 65 , 75
CLINICAL PEARL Pain that is centered immediately in front of the tragus of the ear and that projects to the ear, temple, cheek, and along the mandible is highly diagnostic for TMD. 78 Systems Review
Pain or dysfunction in the orofacial region often results from nonmusculoskeletal causes such as otolaryngologic, neurologic, vascular, neoplastic, psychogenic, and infectious diseases. Clinicians often see patients with a TMD who present with nonspecific symptoms such as neck pain, headaches (see Chapter 5 ), earaches, tinnitus, and sinusitis. 70 Patients with sinusitis complain of acute facial pain or pressure type headaches, and may present with nasal congestion, a reduced sense of smell, postnasal drip, fever or malaise, and aching teeth associated with certain weather conditions or times of the year. 80 Ear disorders, such as an inner or outer ear infection, can produce pre-auricular symptoms in around the TMJ. 81 Conversely, hyperactivity of the masticatory and tensor tympani muscles can cause ear pain, tinnitus, and feelings of fullness in the ear. 82 Finally, patients with eye disorders may experience pain around the eye, numbness, a headache, and other symptoms similar to TMD, a cervicogenic headache (see Chapter 23 ), or a primary headache (see Chapter 5 ). 70 Unexplained weight loss, ataxia, weakness, fever with pain, nystagmus, and neurologic deficits are characteristic of intracranial disorders. 83 Neurovascular disorders are associated with a migraine headache and its variants, carotidynia, and cluster headaches (see Chapter 5 ). Neuropathic disorders include trigeminal neuralgia, glossopharyngeal neuralgia, and occipital neuralgia. A cranial nerve screen (see Chapter 3 ) should be completed on each patient presenting with orofacial pain with particular attention on the fifth cranial nerve (trigeminal), which supplies motor and sensory innervation to the masticatory region. Once the possibility of cervical, systemic, psychogenic, or ear or sinus problems has been ruled out, the next step is to consider the possibility of TMJ pain and impairment. Tests and Measures
Examination of the TMJ structures includes observation, and a thorough mobility and palpation examination to identify impairments and functional limitations. Observation
The posture of the head and neck are assessed for asymmetry. The forward head posture (FHP–see Chapters 6 and 25 ) frequently is associated with TMD. 34 , 84 , 85 This is likely because of the direct impact an FHP can have on oral symmetry during occlusion. Occlusion occurs when the teeth are in contact and the mouth is closed—when tapping the teeth together in the neutral position, all of the teeth appear to strike simultaneously. However, if the same task is attempted while placing the head forward, it is the anterior teeth that occlude first. A deviation from normal occlusion is defined as malocclusion—improper positioning of the teeth and jaws. Malocclusion is a variation of normal growth and development which can affect the bite, the ability to clean teeth properly, gum tissue health, jaw growth, speech development, and appearance. The consequences of this repetitive functional malocclusion during food or gum chewing should be apparent. A chronic FHP may result in an adaptive shortening of the deep cervical fascia and muscles, which can exaggerate the functional malocclusion. Most people have some degree of malocclusion, although it is not usually serious enough to require treatment. Malocclusions can be divided mainly into three types, depending on the sagittal relations of teeth and jaws, by Angle’s classification method, which is based on the relative position of the first maxillary molar 86 : Class I (Neutroclusion): The molar relationship of the occlusion is normal, but the other teeth have problems such as spacing, crowding, and over or under eruption. Class II (Distoclusion): The upper molars are placed anteriorly. There are two subtypes: Class II Division 1 (large overjet): The molar relationships are like that of Class II and the anterior teeth are protruded. Class II Division 2 (deep overbite): The molar relationships are like that of class II but there is lateral flaring of the lateral maxillary incisors. Class III (Mesioclusions): The lower front teeth are more prominent than the upper front teeth. In this case, the patient very often has a large mandible or a short maxillary bone. The clinician should also note whether the teeth are normally aligned or whether there is any crossbite, underbite, or overbite. Crossbite: This occurs when the teeth of the mandible are lateral to the upper maxillary teeth on one side and medial on the opposite side. An anterior crossbite occurs when the lower incisors are anterior to the upper incisors, whereas a posterior crossbite occurs when there is a abnormal transverse relationship of the teeth. Underbite: This occurs when the mandibular teeth are anterior to the maxillary teeth either unilaterally, bilaterally, or in pairs. Overbite: This occurs when the anterior maxillary incisors extend below the anterior mandibular incisors when the jaw is in central occlusion.
Share this:
- Click to share on Twitter (Opens in new window)
- Click to share on Facebook (Opens in new window)
Related posts:

Stay updated, free articles. Join our Telegram channel
Comments are closed for this page.

Full access? Get Clinical Tree

An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
The PMC website is updating on October 15, 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- Angle Orthod
- v.80(1); 2010 Jan

Functional Occlusal Patterns and Their Relationship to Static Occlusion
Kazem s. al-nimri.
a Associate Professor, Department of Orthodontics, School of Dentistry, Jordan University of Science and Technology, Irbid-Jordan
Anwar B. Bataineh
b Professor, Department of Oral and Maxillo-Facial Surgery, School of Dentistry, Jordan University of Science and Technology, Irbid-Jordan
Sawsan Abo-Farha
c Postgraduate student, Department of Orthodontics, School of Dentistry, Jordan University of Science and Technology, Irbid-Jordan
Objectives:
To test the hypothesis that there is no relationship between static occlusion and dynamic occlusion.
Materials and Methods:
The relationship between static and dynamic occlusion was investigated in a sample of 94 dental students (39 males and 55 females) with an age range of 21–30 years. Static occlusion was determined by intraoral examination. Dynamic occlusion was determined in regulated lateral (0.5 mm and 3 mm lateral to the intercuspal position) and protrusive movements of the mandible by intraoral examination with the aid of shimstock.
At the 0.5 mm lateral excursion, 24.5% had bilateral group function and 12.7% had bilateral canine guidance. At the 3 mm positions, the guidance pattern changed to a predominantly canine guidance. Fifty percent of subjects had bilateral canine guidance, and only 8.8% had bilateral group function. In terms of the anterior guidance pattern, a predominant anterior contact with posterior disocclusion (77.5%) was noted. Examination of the relationship between static and dynamic occlusions revealed that at the 0.5 mm position, the pattern of dynamic occlusion was different in relation to various static occlusion features but without reaching a significant level. While at the 3 mm position, the pattern of dynamic occlusion was significantly affected by incisor relationship. The distribution of protrusive excursion patterns was significantly influenced by incisor, canine, and molar relationships.
Conclusions:
The hypothesis is rejected. An association exists between dynamic occlusion and different aspects of static occlusion.
INTRODUCTION
Dental treatment has the capacity to fundamentally change static and dynamic occlusal relationships, while aiming for achieving as near “ideal” occlusion as possible. Accepted criteria for the “ideal” static occlusion were established based on the work of Angle, 1 who is credited with making the profession most aware of occlusion by presenting standards whereby a malocclusion could be compared with normal occlusion, and the work of Andrews, 2 who presented six keys that gave a well-delineated prescription for an ideal intercuspation of teeth. Conversely, the features that constitute “ideal” dynamic occlusion continue to be subject to great debate and have not, to date, been conclusively established.
There are three main concepts regarding tooth contact during the lateral excursion of mandibular movement: (1) balanced occlusion, which was developed from the work of Bonwill, 3 (2) canine guidance, described by D'Amico, 4 and (3) group function, as discussed by Beyron. 5
The relationship between static and dynamic occlusion is one of the aspects of the study of functional occlusion that has received little attention. Few studies have explored the possibility of an association but conflicting results have been reported. Scaife and Holt 6 found canine protected occlusion to be associated with Class II then Class I, and least associated with Class III, malocclusion. The above mentioned findings were confirmed by Al-Hiyasat and Abu-Alhaija 7 in a study of 447 school children, aged 14–17 years. Other studies found that most Class I Angle occlusion cases were associated with balanced occlusion. 8 – 10 On the other hand, Tipton and Rinchuse 11 found no significant association between static and dynamic occlusion.
A number of limitations can be noted in the above mentioned studies: No reference was made to the location of the canine in terms of its relationship to the line of the arch nor to the degree of attrition of the canine, which is of particular importance in examining the assumption that attrition could lead from one type of contact during lateral movement to another. 12 Moreover, the position at which the occlusal contact pattern was recorded (cusp to cusp) is not representative of the functional range of the lateral excursion of mandibular movement.
Another fact for consideration is that changes occurring during occlusal development could influence the occlusal contact pattern; Heikinheimo et al 13 reported an increase in occlusal interferences between the ages of 12 and 15 years in 167 Finns; other studies found a decreasing prevalence with increasing age. 14 , 15 Although the results of these studies are contradictory, they suggest that changes occur during occlusal development that must not be overlooked in sample selection; samples that are beyond the adolescent years would be more representative of a population as it avoids the effects of age and occlusal development on the results of research.
Based on the above mentioned facts, we thought it warranted to further investigate whether a relationship exists between static occlusion and dynamic occlusion, and, if such a relationship exists, which type of dynamic occlusion is associated with which type of static occlusion.
MATERIALS AND METHODS
The population for this study consisted of 94 dental students at Jordan University of Science and Technology (39 males and 55 females). The age of subjects ranged from 21–30 years, with a mean age of 23.1 (±1.44) years. Subjects who met the following criteria were selected:
- No previous or current orthodontic treatment.
- The presence of fully permanent dentition except for the third molars
- No previous occlusal adjustments
- No large restorations involving the incisal edge or a cusp tip
- No crowns or bridges
- No apparent pathologic periodontal problems
- Upper and lower canines in the line of the arch
- No tooth showing attrition into the dentine
The static occlusion of each subject was assessed by intraoral examination on a dental chair under direct vision. The following static occlusal features were recorded:
- Incisor relationship classified according to the British Standard Institute 16
- Canine relationship classified according to Houston et al 16
- Molar relationship classified according to Angle's criteria 1
Dynamic occlusion was determined with the aid of shimstock (Almore shimstock, 8 mm wide, 8 µm thick, Hanel, Langenau, Germany) to confirm tooth contact. The examination was carried out with subjects seated in an upright position in a dental chair with the Frankfort plane parallel to the floor. All recordings were made by the same operator in the same period of the day (morning hours) to avoid possible diurnal variation. 17
For the lateral excursion, occlusal contacts were recorded on the working and the nonworking side at ½ mm and at 3 mm lateral to the habitual centric occlusion. To regulate each lateral position, marks were made on the maxillary central incisor with a water-resistant pencil to mark the intercuspal position at 0.5 mm and 3 mm positions for both right and left sides. Subjects were asked to perform the movements with the aid of a handheld mirror. The shimstock was placed on the occlusal surfaces of teeth from the canine backward; the subject was then asked to close his/her mandible into maximum intercuspation. Gliding movement was performed to the right or the left while the examiner maintained a constant pulling force on the shimstock; on reaching the 0.5 mm position, the teeth holding the shimstock were recorded as working side contacts. The subject was asked to repeat the movement with the shimstock placed on the opposite side to record nonworking side contact. The same procedure was carried out to record occlusal contact at 3 mm lateral to the habitual centric occlusion.
Occlusal contacts at the protrusive excursion of mandibular movement were recorded at the edge-to-edge position. The shimstock was placed on the occlusal surfaces of the anterior teeth; the subject was asked to close into maximum intercuspation and then slide to the edge-to-edge protrusive position while the examiner maintained a constant pulling pressure. Once the teeth were at the edge-to-edge position, teeth holding the shimstock were considered to be in contact and were recorded. The shimstock was then placed on the occlusal surfaces of post teeth, and the subject was asked to repeat the same movement to check for the presence of posterior teeth contact.
In the lateral excursion, the guidance pattern was considered as one of the following:
- Bilateral canine protected occlusion
- Bilateral group function occlusion
- Mixed canine protected and group function
- Bilateral balanced occlusion
- Mixed balanced and group function
- Mixed group function and single tooth contact
- Others (this category included bilateral mediotrusive interference, mixed laterotrusive and canine, bilateral laterotrusive interference, mixed mediotrusive and group, mixed canine guidance, and laterotrusive interference)
In the protrusive excursion, the guidance pattern was considered as one of the following:
- Anterior contact with posterior disocclusion
- Anterior contact with unilateral posterior contact
- Anterior contact with bilateral posterior contact
- No anterior contact with unilateral posterior contact
- No anterior contact with bilateral posterior contact
Ten subjects were reexamined at 2 months following the initial clinical examination to determine intraexaminer reliability. The Kappa ranged from (0.8–1), indicating a reliable examination.
Statistical Analysis
Data were analyzed using the Statistical Package for the Social Sciences (SPSS), version 11 (SPSS Inc, Chicago, Ill). The Chi-square test was used to examine the relationship between the different variables included in this study. P values less than .05 were considered statistically significant.
Dynamic Occlusion
In the total sample at 0.5 mm lateral guidance, 24.5% of subjects had bilateral group function and 18.1% had mixed canine guidance and group function. While at the 3 mm positions, the guidance pattern changed to a predominantly canine guidance. Fifty percent of subjects had bilateral canine guidance, and only 8.5% had bilateral group function ( Table 1 ).
Distribution (%) of Dynamic Occlusion at 0.5 mm and 3 mm Lateral Excursion and Incisor Classification a

In the protrusive guidance patterns, a predominant anterior contact with posterior disocclusion (77.5%) was followed by anterior contact with unilateral posterior contact (12.7%). Anterior contact with bilateral posterior contact was found in 4% of subjects. The remaining 5.8% had no anterior contact with unilateral or bilateral posterior contact.
Static Occlusion
The incisor relationship was Class I in 49 subjects, Class II/1 in 10 subjects, Class II/2 in 17 subjects, and Class III in 18 subjects. The molar and canine relationships are reported in Tables 2 and and3 3 .
Distribution (%) of Dynamic Occlusion at 0.5 mm and 3 mm Lateral Excursion and Canine Classification a

Distribution (%) of Dynamic Occlusion at 0.5 mm and 3 mm Lateral Excursion and Molar Classification a

No statistically significant differences were found between males and females in the various aspects of static occlusion or in the guidance pattern in lateral or protrusive excursions. Therefore, male and female subjects were pooled together in the analysis.
Relationship Between Static and Dynamic Occlusion
The type of guidance at the 0.5 mm position was not significantly associated with the incisor ( Table 1 ), canine ( Table 2 ), or molar classification ( Table 3 ). However, it should be noted that at 0.5 mm lateral excursion, bilateral canine protected occlusion was observed only in subjects with Class I incisor, canine, or molar relationships
As can be seen in Table 1 , 70% of subjects with Class II div 1 and Class II div 2 incisor relationships have bilateral canine protected occlusion at 3 mm lateral guidance, compared with 53% in Class I and 11% in Class III. This difference was statistically significant ( P = .047). On the other hand, no significant association was noted between the type of lateral guidance at the 3 mm positions and the canine relationship ( Table 2 ) or the molar relationship ( Table 3 ).
The pattern of guidance in protrusive mandibular excursions was significantly associated with incisor classification ( P < .0001; Table 4 ), canine classification ( P < .0001; Table 5 ), and molar classification ( P = .031; Table 6 ), with less anterior contact with posterior disocclusion observed in Class III subjects.
Distribution (%) of the Pattern of Guidance in Protrusive Mandibular Excursion and Incisor Classification a

Distribution (%) of the Pattern of Guidance in Protrusive Mandibular Excursion and Canine Classification a

Distribution (%) of the Pattern of Guidance in Protrusive Mandibular Excursion and Molar Classification a

The criteria set for selecting subjects for this study were chosen to ensure the presence of a natural dentition. The absence of apparent pathologic periodontal problems was used as a criterion because the neuromuscular control of occlusion stability and masticatory muscles is influenced by the periodontal afferent. 18 , 19 The fact that the sample was composed of subjects aged 21 to 30 years was chosen because the changes occurring during occlusal development could influence the occlusal contact pattern. Cases with marked attrition were excluded based on the assumption made by McAdam 20 and Woda et al 12 that canine guidance and group function appear to correspond to two successive states of the evolving dentition under the effect of attrition.
Static occlusion was assessed by intraoral examination by direct vision with the aid of a dental mirror, as was done in previous investigations. 7 , 11 Ovsenik et al 21 reported that intraexaminer and inter-examiner malocclusion assessment recorded and measured intraorally is reliable and therefore is proposed as the method of choice to be used not only in epidemiologic studies and screenings, but also in clinical orthodontic assessments.
Occlusal contacts for both lateral and protrusive excursions of the mandible were determined by intraoral examination with the aid of shimstock to confirm the contact between the teeth, as was done in previous investigations. 7 , 22 – 24 Shimstock has been shown to have greater interexaminer reliability than articulating film 25 and high intraexaminer reliability. 7 The chosen thickness of the shimstock was 8 µm, which is below the range of reported thresholds for dental proprioception. This method allows identification of contacting teeth without disturbing this delicate mechanism; a system of measurement that disrupts proprioception may alter mandibular position and consequently tooth contact. 26
For the lateral excursion of the mandible, occlusal contacts were recorded at two positions: ½ mm and 3 mm lateral to the habitual centric. These two positions were selected based on the findings of Ogawa and coworkers, 23 who conducted a study in which tooth contacts were recorded at 0.5, 1, 2, and 3 mm lateral to the maximum intercuspation. They concluded that the occlusal contact patterns during lateral movement varied greatly with mandibular position. The occlusal contact pattern in the 3 mm position predicted the presence or absence of the occlusal contact in the 1 and 2 mm positions (sensitivity >0.7) but not in the 0.5 mm position (sensitivity <0.6). Finally, they suggest that the 0.5 mm position could be used to evaluate occlusal contact in a position close to maximum intercuspation in the functional range, and that the 3 mm position could be used to assess occlusal contact in an edge-to-edge position in the parafunctional range. 24
The orthodontic picture of “ideal occlusion” places considerable emphasis on the static occlusal relationship in assessing the quality of completed orthodontic treatment, with less emphasis on the importance of the dynamic occlusion. In fact, none of the available orthodontic indices for the assessment of treatment outcome contains any functional components! One could argue that this is caused by the absence of consensus regarding what constitutes an “ideal” dynamic occlusion. This, nonetheless, should not lead to a practice of disregarding basic functional principles during orthodontic treatment. As Clark and Evans 27 argue, the gradual adaptation of muscles and joints that occurs during the slow development of a specific occlusion during growth may not occur following the much quicker change related to orthodontic treatment. Other possible consequences of occlusal interference, such as tooth wear and relapse of tooth position, may become apparent only some time after completion of orthodontic treatment, but nevertheless may be attributable to interferences introduced during appliance therapy.
Based on all of the above, we found it particularly interesting to explore the relationship between static and dynamic occlusion. At 0.5 mm lateral excursion, canine protected occlusion was more dominant in Class I incisor, canine, and molar relationships; at 3 mm lateral excursion, canine protected occlusion was dominant in Class II occlusion. Our findings at the 3 mm position compare favorably with those of Al-Hiyasat and Abu-Alhaija, 7 who reported that canine guidance was more dominant in Class II followed by Class I; and those of Scaife and Holt, 6 who found canine protected occlusion to be associated with Class II then Class I and least associated with Class III. However, these results do not agree with other studies, in which investigators found most of Class I Angle occlusion to be associated with balanced occlusion 8 – 10 ; neither do they agree with the findings of Tipton and Rinchuse, 11 who found no significant association between static and dynamic occlusion.
This lack of agreement may be attributed to the different registration materials utilized. Registration material has a significant influence on the number of contacts recorded; some materials tend to record “near contact” as actual contact resulting in differences in the occlusal contact pattern registered. 28 Diurnal difference in occlusal contacts, 17 differences in the criteria used for sample selection, and differences in the classification system are additional factors that contribute to the observed differences.
Anterior guidance with posterior distocclusion was associated with Class II div 2 incisors, followed by Class I and Class II div 1; Class II canines followed by Class I; and Class II molars followed by Class I. These associations explain the high prevalence of posterior contact in protrusion observed in Class III cases and the high prevalence of anterior contact with posterior distocclusion in Class II div 2 cases.
These observations are consistent with those of Al-Hiyasat and Abu-Alhaija, 7 who reported that anterior guidance with posterior distocclusion was associated with Class II div 2 incisors, then Class I and Class II div 1. The prevalence of posterior contact in protrusion was dominantly associated with Class III incisor and molar relationships (50% and 33%, respectively).
Although an association between static and dynamic occlusion was found, it is very difficult to establish a definitive association between them. Therefore, it is necessary to evaluate dynamic occlusion with the aspects of length and inclination of the occlusal guidance of each tooth used as functional indicators.
CONCLUSIONS
- The distribution of lateral guidance is different at the 0.5 mm and 3 mm positions.
- At 0.5 mm lateral excursion, bilateral canine protected occlusion was observed only in subjects with Class I incisor relationships or subjects with unilateral or bilateral Class I molar or canine relationships.
- At 3 mm lateral excursion, bilateral canine protected occlusion was predominant in subjects with Class II incisor, canine, and molar relationships.
- Anterior guidance with posterior disocclusion was observed most often in subjects with Class II div 2 occlusion and was least observed in subjects with Class III occlusion.
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- My Account Login
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- Open access
- Published: 19 September 2024
N-acetylaspartate promotes glycolytic-to-oxidative fiber-type switch and resistance to atrophic stimuli in myotubes
- Serena Castelli ORCID: orcid.org/0000-0001-5857-8049 1 ,
- Enrico Desideri 2 ,
- Leonardo Laureti 3 ,
- Federica Felice ORCID: orcid.org/0000-0001-8250-4680 3 ,
- Angela De Cristofaro 3 ,
- Silvia Scaricamazza 4 ,
- Giacomo Lazzarino 5 ,
- Maria Rosa Ciriolo ORCID: orcid.org/0000-0002-7863-9029 1 , 3 na1 &
- Fabio Ciccarone ORCID: orcid.org/0000-0002-2425-2240 1 , 3 na1
Cell Death & Disease volume 15 , Article number: 686 ( 2024 ) Cite this article
Metrics details
- Biochemistry
- Neuroscience
N-acetylaspartate (NAA) is a neuronal metabolite that can be extruded in extracellular fluids and whose blood concentration increases in several neurodegenerative disorders, including amyotrophic lateral sclerosis (ALS). Aspartoacylase (ASPA) is the enzyme responsible for NAA breakdown. It is abundantly expressed in skeletal muscle and most other human tissues, but the role of NAA catabolism in the periphery is largely neglected. Here we demonstrate that NAA treatment of differentiated C2C12 muscle cells increases lipid turnover, mitochondrial biogenesis and oxidative metabolism at the expense of glycolysis. These effects were ascribed to NAA catabolism, as CRISPR/Cas9 ASPA KO cells are insensitive to NAA administration. Moreover, the metabolic switch induced by NAA was associated with an augmented resistance to atrophic stimuli. Consistently with in vitro results, SOD1-G93A ALS mice show an increase in ASPA levels in those muscles undergoing the glycolytic to oxidative switch during the disease course. The impact of NAA on the metabolism and resistance capability of myotubes supports a role for this metabolite in the phenotypical adaptations of skeletal muscle in neuromuscular disorders.
Introduction
N-acetylaspartate (NAA) is the second most abundant metabolite in the central nervous system (CNS) reaching concentrations of approximately 10 mM or greater. NAA is synthesized from aspartate and acetyl-CoA by the mitochondrial enzyme N-acetyltransferase 8 like (NAT8L), whereas the catabolism is due to aspartoacylase (ASPA) enzyme activity. In the CNS, NAA synthesis mainly occurs in neurons, which accumulate it due to the almost complete lack of ASPA [ 1 ]. NAA can be released in the CNS extracellular space and promptly taken up by glial cells, particularly oligodendrocytes, which express ASPA enzyme and can utilize NAA as a source of acetyl-CoA for fatty acid synthesis and acetylation processes [ 2 ]. Given that NAA is predominantly stored inside neuronal cells, NAA concentration in cerebrospinal fluid and blood is generally low in healthy subjects, but it significantly increases in neurodegenerative diseases [ 3 ]. Despite the ability of NAA to reach any district of the body via the circulatory system, few studies have focused on the role of the NAA pathway in peripheral tissues. Among them, NAA metabolism in tissues other than the brain has been assessed in brown adipose tissue (BAT). Brown adipocytes express NAT8L enzyme at levels almost comparable to those of neurons and, expressing also ASPA enzyme, they can simultaneously break down NAA [ 4 , 5 ]. In this tissue, NAA synthesis and catabolism are coupled with an increase in lipogenesis and lipolysis, determining a “futile” cycle that continuously supports mitochondrial oxidation and thermogenesis [ 6 , 7 ]. Apart from neurons and brown adipocytes, NAT8L expression is very weak in other organs while ASPA expression is more widely distributed in all peripheral tissues, including skeletal muscle, suggesting that NAA catabolism has functional consequences in the periphery.
Skeletal muscle, the most abundant tissue of the human body, displays heterogeneity in myofibers that depends on the speed of contraction and the energetic fuel they use. Myofibers can be divided into two major types, type I (or slow-twitch) fibers and type II (or fast-twitch) fibers. Type II fibers can be further divided into the subtypes IIA and IIB/X [ 8 ]. The slow-twitch type I myofibers have a slow speed of contraction and are the most fatigue-resistant. They are highly vascularized and show high levels of mitochondria and myoglobin, and mostly rely on lipid catabolism and oxidative phosphorylation for energy production. The fast-twitch type IIB/X fibers are characterized by a rapid contraction of the sarcomere, are easily fatigued, and mainly depend on glycolysis for ATP production; the type IIA fibers exhibit an intermediate behavior and metabolism between slow-twitch type I fibers and highly glycolytic type IIB/X fibers. The relative abundance of each type of fibers in muscles is influenced by several factors like lifestyle, training, age and pathological conditions [ 6 ]. Notably, glycolytic-to-oxidative fiber-type switch has been observed in motor neuron diseases such as amyotrophic lateral sclerosis (ALS), where muscles undergo an increase in lipid oxidative metabolism and replace type-IIB myosin heavy chain (MHC) isoforms with type IIA ones [ 9 , 10 ].
Considering the metabolic alterations occurring in ALS muscle, the fact that serum NAA levels increase in ALS [ 11 ] and that the degeneration of spinal motor neurons may locally increase NAA concentration, we aimed to characterize the contribution of NAA in the rewiring of muscle cell metabolism. We analyzed the effects of NAA treatment in an in vitro model of skeletal muscle, namely differentiated C2C12 myotubes. Metabolic consequences of NAA administration were coupled with analysis of myotube morphology, identity, and response to atrophic stimuli. Evidence of a key role of NAA catabolism on muscle metabolism was provided by using ASPA CRISPR/Cas9 knock-out (KO) C2C12 cells, published datasets and muscle biopsies from the SOD1-G93A ALS mouse model.
Materials and methods
Materials used for this work are detailed in the Supplementary Materials.
Cell lines, treatments and animals
The cell line of murine myoblasts C2C12 was purchased from the American Type Culture Collection (ATCC). Cells were grown in Dulbecco’s modified Eagle’s medium (DMEM) 4.5 g/L glucose, supplemented with 10% fetal bovine serum, 10 U/ml penicillin/streptomycin, 2 mM L-glutamine, 1 mM sodium pyruvate and 10 mM HEPES pH 7.0. Cells were cultured at 37 °C in an atmosphere of 5% CO 2 in the air and, for all the experiments, they were plated at a 5 × 10 4 cells/mL density on wells covered by 0.1 g/mL of gelatin derived from bovine skin (G9391, Sigma Aldrich). After 24 h, C2C12 differentiation in myotubes was induced by a specific differentiation medium consisting of DMEM containing 4.5 g/L of glucose, supplemented with 2% of horse serum (HS), 2 mM of L-glutamine, 10 U/mL penicillin/streptomycin solution, 1 mM sodium pyruvate and 10 mM HEPES pH 7.0. The differentiation was induced for 7 days, changing the medium every day, for all experiments, except where differently indicated. The treatment with NAA (Sigma Aldrich, 00920) was added after 5 days of differentiation and was left for 48 h, until the end of differentiation.
SOD1-G93A mice (B6.Cg-Tg(SOD1 G93A)1Gur/J) were purchased from The Jackson Laboratory (Bar Harbor, ME, USA). Hemizygous SOD1-G93A males and C57BL/6 females were crossbred, and progeny were genotyped by PCR. Disease onset was evaluated by hanging grid test; 20–22 weeks-old mice were defined as “symptomatic”. Before sacrifice, mice were anaesthetized with Rompum (xylazine, 20 mg/ml, 0.5 ml/kg Bayer, Milan, Italy) plus Zoletil (tiletamine and zolazepam, 100 mg/ml, 0.5 ml/kg; Virbac, Milan, Italy). A detailed description of treatments and animals’ management was provided in Supplementary Materials.
Knock-out production by CRISPR/Cas9 technology
The generation of ASPA and ATGL Knock-Out (KO) C2C12 cells was performed by the CRISPR/Cas9 technology. For ASPA KO, the Cas9 enzyme was directed by gRNA, obtained from the following guides: ASPA 1, forward 5'-CACCGAGTGCAACCCATGTTAGAAG-3' and reverse 5'-AAACCTTCTAACATGGGTTGCACTC-3'; ASPA 2 forward 5'-CACCGAGTGCAACCCATGTTAGAAG-3' and reverse 5'-AAACCTTCTAACATGGGTTGCACTC-3'. For ATGL KO, the following guides were used: ATGL 1, forward 5'-CACCGAGAGGCGGTAGAGATTGCGA-3' and reverse 5'-AAACTCGCAATCTCTACCGCCTCTC-3'; ATGL 2, forward 5'-CACCGGCAGGAGGCCACGCCAATG-3' and reverse 5'-AAACCATTGGCGTGGCCTCCTGCC-3'. For control, only the Cas9 plasmid was transfected. The detailed construction was provided in Supplementary information.
Western blot analysis
Detailed methods and antibodies were provided in Supplementary information.
Fluorescence microscopy analyses
Live cells were treated with 2.5 µM JC-1 (420200, Calbiochem) for 30 min at 37 °C. Delta Vision Restoration Microscopy System (Applied Precision, Issaquah, WA) equipped with an Olympus IX70 fluorescence microscope (Olympus Italia, Segrate, Milano, Italy) was used to acquire fluorescent images of cells. Detailed methods were provided in Supplementary information.
Extracellular lactate assay
Cell medium was precipitated with 30% trichloroacetic acid (TCA). Media were stored at −20 °C for at least 1 h and then centrifuged at 14,000 × g for 20 min at 4 °C. Supernatant was incubated with reaction buffer containing LDH enzyme for 30 min at 37 °C. The amount of lactate was quantified by measuring NADH absorbance at 340 nm using an Eppendorf BioSpectrometer. Concentrations were normalized on total proteins in each sample. The detailed protocol was provided in Supplementary Materials.
ATP quantification assay
ATP quantification was performed by using ATP Assay Kit - Colorimetric Kit (Assaygenie). The assay protocol relies on the production of creatine phosphate from creatine and ATP. The content of phosphocreatine is detected by colorimetric method to reflect ATP content.
Oil Red-O staining
Cells were fixed in formalin solution neutral buffered 10% and, after PBS washes, were incubated for 5 min with 60% isopropanol and stained with the Oil Red-O for 10 min. Oil Red-O was removed and di-deionized water was used to wash cells. Delta Vision Restoration Microscopy System (Applied Precision) equipped with an Olympus IX70 fluorescence microscope (Olympus) was used to acquire fluorescent images of cells. Detailed methods were provided in Supplementary information.
Complex I activity assay
The activity of the NADH:ubiquinone oxidoreductase complex I was measured through a direct method [ 12 ], following the decrease in absorbance at 340 nm due to the oxidation of NADH. A detailed description of this protocol is provided in Supplementary Materials.
RNA isolation and real-time qPCR
RNA extraction was performed by using TRItidy G (PanReac AppliChem). Synthesis of cDNA was obtained from 1 μg of total RNA by using iScript™ Reverse Transcription Supermix for RT-qPCR (Bio-Rad), and RT-qPCR reaction was performed by using the iTaq Universal SYBR Green Supermix (Bio-Rad) on QuantStudio™ 3 real-time PCR System (Thermo Fisher Scientific). The list of primers is contained in Supplementary Materials.
HPLC analysis
For HPLC analysis, details were provided in Supplementary Materials.
Bioinformatic analyses
The detailed list of databases used was provided in Supplementary Materials.
NAA promotes lipid turnover and an oxidative phenotype in C2C12 myotubes
To identify the effects of NAA on the metabolism of diffeentiated C2C12 myotubes, NAA was administered at day 5 of differentiation and maintained for 48 h (Fig. 1A ). At that time, cells showed the highest expression of the late differentiation marker MHC (Supplementary Fig. 1A, B ) and myotubes were already formed (Supplementary Fig. 1C ).
A Graphical summary of the experimental design. B Western blot analysis of acetyl-lysine levels. β-Actin was used as a loading control. C Representative images of differentiated C2C12 myotubes after staining with Oil Red-O. Scale bars, 50 µm. (n = 3) D The ratio of MFI of untreated and treated cells with ATGListatin. Data are expressed as mean ± SD of n = 3 independent experiments (*** p < 0.001 vs untreated). E Colorimetric quantification of ATP after 2 mM NAA treatment and co-treatment with oligomycin (2 µM), 2-deoxyglucose (40 mM) and etomoxir (20 µM). Data are expressed as mmol of ATP produced per µg of protein. ( ** p < 0.01¸ *** p < 0.001). F Western blot analysis of PGC1α, NRF1, ACO2 and TFAM levels on C2C12 myotubes. β-Actin was used as loading control. The Western blots reported are representative of three independent experiments that gave similar results. G Evaluation of extracellular lactate content after NAA co-treatment with ATGListatin. Lactate concentration was normalized on total proteins and data are expressed as mean ± SD of n = 3 independent experiments ( ** p < 0.01 vs 0 mM). RT-qPCR analysis of H MyH4, MyH2 , MyH7b and I myoglobin in C2C12 myotubes. ACTB was used as a reference control. Data are shown as fold change ± SD of n = 3 independent experiments (* p < 0.05, ** p < 0.01 vs CTRL). CTRL was represented by a straight line in the bar graph.
NAA is known to provide acetyl-CoA for acetylation processes and fatty acid synthesis [ 13 ], thus we examined acetyl-lysine levels and lipid droplets (LDs) content. As expected, acetyl-lysine levels increased after NAA treatment (Fig. 1B ; Supplementary Fig. 1D ). Although Oil Red-O staining showed a decrease in LDs content after NAA administration, in myotubes co-treated with the lipolytic inhibitor ATGListatin or knock-out (KO) for Adipose triglyceride lipase (ATGL) the accumulation of LDs was higher than control cells (Fig. 1C, D ; Supplementary Fig. 1E-H ). This result is compatible with a boosted lipid synthesis and degradation in these cells. The increased lipogenesis was further confirmed by the dephosphorylation/activation of Acetyl-CoA carboxylase1 (ACC1) (Supplementary Fig. 1I, J ) while the increase in lipid catabolism following NAA administration was confirmed by the decrement of Oil Red-O staining in cells pre-treated with oleic acid (Supplementary Fig. 1K ), which was used for boosting LDs accumulation, and by the upregulation of the lipases ATGL and HSL (Supplementary Fig. 1L, M ). Confirmatory results of the effect of NAA on LDs turnover were obtained both with higher concentrations of NAA (Supplementary Fig. 2A, B ), and when NAA treatment was performed on C2C12 cells differentiated for 15 days (Supplementary Fig. 2C, D ).
This augmentation in lipid catabolism was also corroborated by the increase in ATP production after NAA treatment, which was dramatically abrogated by blocking mitochondrial fatty acid import with etomoxir (Cpt1α inhibitor). This results indicates that the increase in ATP levels after NAA treatment is mainly due to oxidative phosphorylation fueled by fatty acid oxidation (Fig. 1E ) as confirmed by the decreased ATP concentration observed after oligomycin (ATP synthase inhibitor) treatment and not after inhibition of glycolysis by 2-deoxyglucose (2-DG). Coherently, mitochondrial mass was also augmented by NAA, as shown by the increase of ACO2 and TFAM levels, thanks to a prompted mitochondrial biogenesis, as suggested by higher levels of PGC1α and NRF1 (Fig. 1F ; Supplementary Fig. 2E ). The increase in mitochondrial membrane potential revealed by the increased ratio between the red and green fluorescence of the JC-1 dye (Supplementary Fig. 2F, G ) and the high activity of mitochondrial complex I clarified that mitochondria are more active after NAA treatment (Supplementary Fig. 2H ). This increase in mitochondrial oxidative metabolism was paralleled by a decrease in the glycolytic rate as demonstrated by the reduction of the extracellular lactate extruded by NAA-treated C2C12 myotubes (Fig. 1G ). Notably, this metabolic shift was likely due to the higher lipid usage induced by NAA, as blocking lipid catabolism by ATGListatin did not alter extracellular lactate levels in NAA-treated cells (Fig. 1G ). As myofiber identity corresponds to a specific metabolic setting [ 6 , 14 ], the markers of the three types of myofibers (slow- intermediate- and fast-twitch) were evaluated after NAA treatment. MHC IIb (encoded by MyH4 gene), typically expressed in fast glycolytic fibers, was decreased, whereas the fast oxidative/intermediate and slow oxidative markers MHC IIa (encoded by MyH2 gene) and MHC7b (encoded by MyH7b gene) were increased by NAA (Fig. 1H ). These results demonstrate that NAA elicits a glycolytic-to-oxidative fiber-type switch in muscle cells, as further confirmed by the upregulation of myoglobin (Fig. 1I ), which supports oxidative phosphorylation in oxidative myofibers.
NAA decreases myofiber diameter by enhancing protein turnover
In order to evaluate whether the molecular and metabolic signatures induced by NAA on C2C12 myotubes are associated with phenotypic changes in the fibers, we measured myotube diameters in brightfield images. We observed that NAA treatment reduced the diameter of myotubes (Fig. 2A , B), which is consistent with the acquisition of slow-twitch fiber phenotypes [ 8 ]. The different diameter between NAA-treated and untreated myotubes was not due to a discrepancy in the differentiation capability or the activation of atrophic response after NAA treatment, as demonstrated by the unchanged fusion index (Supplementary Fig. 3A ) and the unaltered or even reduced levels of the main atrophic markers TRIM63 (also known as Murf1) and FBXO32 (also known as Atrogin1) (Supplementary Fig. 3B ).
A Bright-field images of C2C12 myotubes. Scale bars, 50 µm. (n = 3). B The diameter measurement performed by ImageJ software. Data are expressed as mean ± SD of n = 3 independent experiments (* p < 0.05 vs 0 mM). C Western blot analysis of p-mTOR, mTOR and p-p70s6k (Thr389) levels in C2C12 myotubes. β-Actin was used as a loading control. D Western blot analysis of puromycin incorporation levels. Ponceau S was used as a loading control. Western blot analysis of E ubiquitinated protein levels and F BNIP3 and LC3 levels with/without NH 4 Cl and leupeptin. β-Actin was used as a loading control. The Western blots reported are representative of three independent experiments that gave similar results. G The diameter measurement of bright-field images of myotubes MG132 or NH 4 Cl/leupeptin treatment was performed by ImageJ software. Data are expressed as mean ± SD of n = 3 independent experiments (* p < 0.05, ** p < 0.01 vs untreated).
Thereafter, we evaluated the main pathways involved in protein synthesis and degradation that can promote a higher protein turnover rate, which is a feature of type I fibers and a process required for myofiber remodeling [ 15 ]. The higher phosphorylation levels of mTOR-S6K pathway components demonstrated a more active protein synthesis (Fig. 2C ; Supplementary Fig. 3C, D ) in C2C12 cells treated with NAA, as also confirmed by the puromycin incorporation assay (Fig. 2D , Supplementary Fig. 3E ). The proteasomal degradation pathway was analyzed by monitoring the levels of ubiquitinated proteins, which resulted slightly increased after NAA treatment (Fig. 2E ; Supplementary Fig. 3F ). The high levels of BNIP3 and LC3-II in NAA-treated cells upon autophagy inhibition with NH 4 Cl/leupeptin demonstrated a massive activation of autophagy after NAA treatment (Fig. 2F ; Supplementary Fig. 3G, H ). Given the activation of both these degradation pathways upon NAA treatment, their role in myotube phenotypic changes was tested. By blocking either proteasome or autophagy, the reduction in myotube diameter was abrogated upon NAA (Fig. 2G , Supplementary Fig. 3I ).
The metabolic and phenotypical effects of NAA are determined by its catabolism
To clarify the role of NAA catabolism in the metabolism of myotubes, we first characterized the expression of ASPA enzyme in the skeletal muscle. ASPA expression was comparable to that of the majority of human peripheral tissues, except adipose tissue and kidney, which exhibited very high expression levels [ 16 ] (Supplementary Fig. 4A ). ASPA levels were higher in the oxidative muscle soleus than in the glycolytic muscle extensor digitorum longus (EDL) (Fig. 3A ). Gene ontology (GO) analysis of genes that correlate with ASPA expression in muscle identified the GO terms linked to oxidative phosphorylation and fatty acid oxidation as the most significant ones (Fig. 3B ). These results demonstrated a strong association between ASPA and oxidative metabolism in myofibers. Consistently, ASPA was upregulated during the glycolytic-to-oxidative switch we obtained after NAA treatment (Fig. 3C, D ; Supplementary Fig. 4B ).
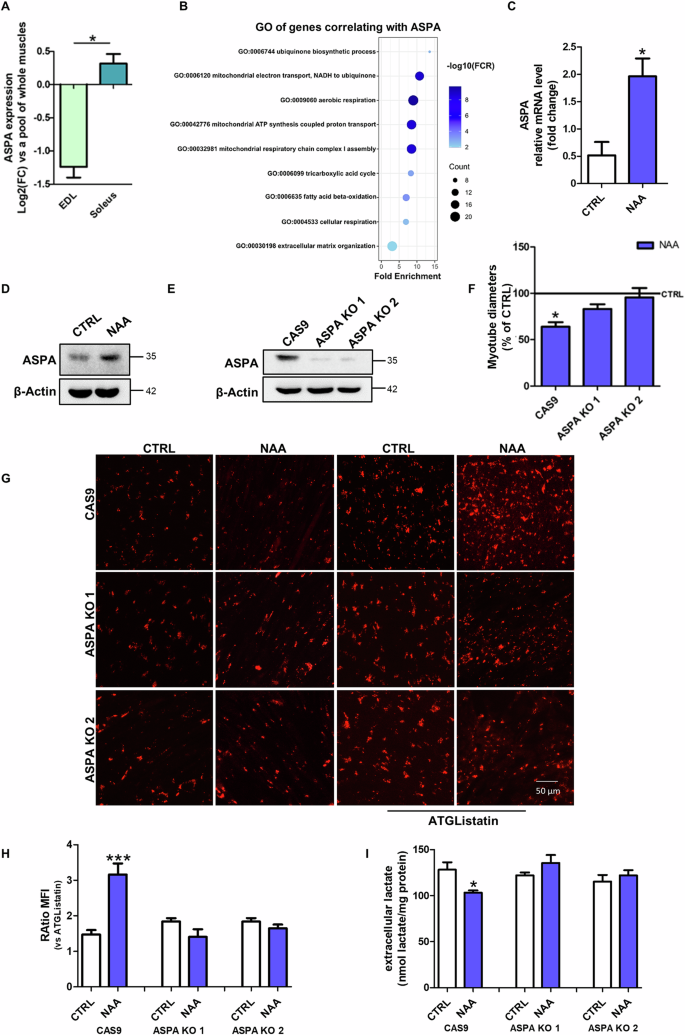
A Bioinformatic analysis of ASPA expression in EDL and soleus muscles. Data are expressed as Log 2 (fold change) ± SD ( *p < 0.05 EDL vs soleus). B Bioinformatic analysis of GO of transcripts that correlate with ASPA expression in muscle. C RT-qPCR analysis of ASPA expression in C2C12 cells. ACTB was used as a reference control. Data are shown as fold change ± SD of n = 3 independent experiments (* p < 0.05 vs CTRL). Western blot analysis of ASPA levels on D C2C12 myotubes treated with NAA for 48 h and on E differentiated ASPA KO C2C12 cells. The Western blots reported are representative of three independent experiments that gave similar results. F Diameter measurement of C2C12 ASPA KO cells treated with NAA. Data are expressed as mean ± SD of n = 3 independent experiments ( *p < 0.05 vs CTRL). G Representative images of C2C12 ASPA KO myotubes after staining with Oil Red O. Scale bars, 50 µm. (n = 3). H The ratio of MFI of cells untreated and treated with ATGListatin. Data are expressed as mean ± SD of n = 3 independent experiments (*** p < 0.001 vs untreated). I Evaluation of extracellular lactate content in C2C12 ASPA KO cells. Lactate concentration was normalized on total proteins and data are expressed as mean ± SD of n = 3 independent experiments (* p < 0.05 vs CTRL Cas9).
Based on this evidence and to investigate whether NAA catabolism is required for the effects elicited by NAA treatment we generated ASPA KO C2C12 cells using CRISPR/Cas9 technique (Fig. 3E ; Supplementary Fig. 4C ). ASPA KO prevented NAA-induced changes in myotube diameter (Fig. 3F ; Supplementary Fig. 4D ), LDs content (Fig. 3G, H ) and lactate extrusion (Fig. 3I ). These results demonstrated that the phenotypic and metabolic effects of NAA were dependent on its catabolism by ASPA.
Myotubes treated with NAA are more resistant to atrophic stimuli
Oxidative fibers are known to be more resistant not only to physiological stimuli, such as exercise and fatigue, but also to atrophy [ 17 ]. Therefore, we monitored the response of NAA-treated myotubes challenged with atrophic stimuli. The pro-atrophic stimulus dexamethasone (DEXA) decreased the diameter of control cells, but not NAA-treated myotubes (Fig. 4A , B). Similar results were obtained using two additional atrophic stimuli, TNFα and cisplatin (Supplementary Fig. 4E, F ).
C2C12 myotubes were treated with dexamethasone (DEXA) and after 48 h ( A ) bright field images of myotubes were taken. Scale bars, 50 µm (n = 3). B The diameter measurement was performed by ImageJ software. Data are expressed as mean ± SD of n = 3 independent experiments ( *** p < 0.001 vs % of untreated). C Bioinformatic analysis of ASPA expression in gastrocnemius muscle of WT, SOD1-G93A and nerve-crushed mice. Data are expressed as Log2(fold change) ± SD ( *p < 0.05 vs WT ). Western blot analysis of ASPA, HSL and ATGL levels on ( D , F ) quadriceps muscle and ( E , G ) gastrocnemius muscle of symptomatic SOD1-G93A and WT mice. β-Actin was used as a loading control. Determination of NAA concentration in H serum, I spinal cord and J BAT of SOD1-G93A and WT mice by HPLC. K Western blot analysis of ASPA on brown adipose tissue of symptomatic SOD1-G93A and WT mice. β-Actin was used as a loading control. The Western blots reported are representative of three independent experiments that gave similar results.
As NAA catabolism was shown to be fundamental for the metabolic changes that lead to the glycolytic-to-oxidative fiber switch, we verified whether ASPA expression was modulated in in vivo models characterized by muscle atrophy. We observed that ASPA levels were increased in the gastrocnemius muscle of asymptomatic SOD1-G93A ALS mice and wild-type sciatic nerve-crushed mice, reaching statistical significance in this latter group (Fig. 4C ). We then experimentally demonstrated the protein levels of ASPA were significantly upregulated in quadriceps and gastrocnemius of symptomatic SOD1-G93A ALS mice (Fig. 4D, E ; Supplementary Fig. 5A, C ) in parallel with high levels of the ATGL and HSL lipases (Fig. 4F, G ; Supplementary Fig. 5B, D ), which are consistent with the well-known metabolic switch from glycolytic to oxidative fibers in these muscles during ALS disease [ 18 , 19 ].
ALS pathology is characterized by motor neuron degeneration and systemic hypermetabolism that we observed to be associated with BAT overactivation [ 20 ]. As these are the tissues that express the highest levels of the NAA-producing enzyme NAT8L, we extended our analysis on the NAA pathway to the spinal cord and BAT to characterize the sources of NAA that can feed muscles in SOD1-G93A ALS mice. After validating that serum NAA levels were increased in ALS mice (Fig. 4H ), as previously described in humans, we demonstrated that this condition was coupled with a decreased content of the metabolite in the spinal cord (Fig. 4I ) and no change in ASPA levels (Supplementary Fig. 5E, F ). On the contrary, NAA was higher than controls in BAT of SOD1-G93A mice (Fig. 4J ), whereas ASPA was decreased (Fig. 4K ; Supplementary Fig. 5G ).
This study highlighted for the first time the role of NAA catabolism in the metabolic adaptation of skeletal muscle. NAA administration augments lipid turnover in differentiated C2C12 myotubes, promoting both lipid synthesis and catabolism, as also observed in brown adipocytes overexpressing NAT8L [ 13 ]. NAA catabolism is necessary for the stimulation of lipid turnover, as no significant changes are detected in ASPA KO myotubes. The effect on lipogenesis is reasonably caused by the increase in acetate levels that can directly provide cytosolic acetyl-CoA as a substrate for the ACC enzyme. As concerns lipolysis, its activation may be the consequence of LDs accumulation to avoid that muscle cells may abnormally accumulate neutral lipids, a condition that is known to be detrimental for the muscle as demonstrated in several genetic diseases defective for lipid catabolic pathways [ 21 ]. We can also hypothesize that acetate itself may contribute to the activation of lipid catabolism as the enhancement of lipolysis was demonstrated in the skeletal muscle of mice fed with a supplementation of acetate [ 22 ]. A direct signaling role of NAA per se in both lipogenesis and lipolysis cannot be excluded.
The increase in lipid utilization is consistently supported by the stimulation of mitochondrial activity and biogenesis in C2C12 myotubes. Together these data and the upregulation of myoglobin demonstrated that the treatment with NAA promotes an oxidative phenotype in differentiated myotubes. Notably, this induction in the oxidative pathway was coupled to a decrease in anaerobic glycolysis after NAA treatment. This result was reverted when a lipolysis inhibitor or ASPA KO cells were used, further demonstrating that NAA catabolism and consequent ATGL activation are the drivers of the metabolic shift boosting mitochondrial metabolism while inactivating glycolytic flux.
Besides metabolism, NAA treatment promoted other phenotypic changes typical of oxidative myofibers, including the upregulation of MHC7b and MHC IIa and the increase in protein turnover (increased protein synthesis and autophagic/proteasomal degradation). Consistently, the glycolytic-to oxidative switch described in ALS skeletal muscle [ 10 ] and in the spinal and bulbar muscular atrophy [ 23 ], is also associated with the upregulation of oxidative-specific MHC. On the contrary, this change in MHC isotypes was not observed when the oxidative muscle soleus shifted towards a glycolytic phenotype in mice defective for mitochondrial long-chain fatty acid oxidation [ 24 ].
In this scenario, the upregulation of PGC1α following NAA treatment seems to be fundamental as it is the master player in the glycolytic-to-oxidative switch of muscle cells promoting mitochondrial biogenesis, oxidative metabolism and the expression of type I MHC isoforms [ 25 , 26 ]. Moreover, PGC1α is also known to protect skeletal muscle from atrophy, indirectly, by buffering ROS [ 27 ] and, directly, by suppressing atrophy-specific gene transcription [ 17 ]. This is consistent with the slight reduction of FBXO32 expression after NAA treatment and the inhibitory action of NAA on the diameter of myotubes challenged with different atrophic stimuli.
Beyond ALS, the glycolytic-to-oxidative switch of the skeletal muscle is frequently associated with muscle atrophy also in other physiological and pathological conditions. Age-associated skeletal muscle atrophy in glycolytic fibers is linked to a decrease in mitochondrial enzyme expression while type I fibers seem to be initially protected due to the higher content of mitochondria [ 19 ]. Analogously, the decrease of PGC1α expression and oxidative fiber content has a detrimental effect on muscle homeostasis leading to muscle atrophy in Huntington’s disease [ 28 ]. Conversely, oxidative myofibers are largely exposed to atrophy after muscle disuse and shift toward glycolysis [ 29 ]. Therefore, the metabolic changes occurring in such conditions are likely to be an adaptation to limit skeletal muscle dysfunction, most likely because oxidative fibers are more resistant than glycolytic ones. In the context of pathological conditions linked to motor neuron damage such as ALS, in which NAA accumulation occurs in blood, the oxidative phenotype promoted by NAA treatment and the inhibition of atrophic response suggest that NAA functions as a protection/compensation mechanism, forcing ALS muscles towards a more resistant oxidative phenotype. This is also supported by the upregulation of ASPA enzyme we described in ALS muscles undergoing the glycolytic-to-oxidative switch and in denervated muscles.
An important aspect to consider is the source of NAA that can impact skeletal muscle metabolism. Differently from neurons and brown adipocytes, muscle cells show very low or no expression of NAT8L. A major interaction between NAA and skeletal muscle can occur in those pathological states showing increased blood concentration of NAA and muscle alterations, including ALS. A direct release of NAA at the level of the neuromuscular junction can occur following the degeneration of motoneurons. We cannot exclude that local production of NAA in muscle can also derive from immune cells that can be present in both homeostatic and injured skeletal muscle as well as from the muscle fibro-adipogenic progenitors that can transdifferentiate in brown/beige adipocytes to recover muscle degeneration [ 30 ]. In the SOD1-G93A mouse model, we observed an increase of NAA levels in serum while a reduction in the spinal cord that is consistent with the degenerative process of the tissue. Therefore, neurodegeneration is likely to represent the primary mechanisms involved in NAA release due to the very high abundance of this metabolite in neurons. Nevertheless, the well-known cross-talk between BAT and skeletal muscle has to be also mentioned given that a hyperactivation of BAT in ALS mouse models was demonstrated [ 20 ]. The increased production of NAA in ALS BAT, which is coupled with its impaired intracellular catabolism due to ASPA downregulation, may partially contribute to NAA release during the disease course as a consequence of muscle metabolic reprogramming.
Overall, our results highlighted an unpredicted role of NAA as a key driver of muscle metabolic identity that, fostering lipid turnover and oxidative metabolism, may safeguard myotubes from physiological or disease-associated atrophic conditions.
Data availability
Bioinformatic analyses of ASPA expression in EDL and soleus muscle were performed on microarray data and the raw data is available in the GEO database (accession number GSE23244). The expression levels of ASPA in gastrocnemius of SOD1-G93A and nerve-crushed mice are available from the GSE16362 dataset. ASPA expression in the different tissues was analyzed from the Genotype-Tissue Expression dataset (GTex; https://www.gtexportal.org ). Gene ontology (GO) enrichment analysis of genes co-expressed with ASPA has been performed using the Database for Annotation, Visualization and Integrated Discovery (DAVID) web tool ( https://david.ncifcrf.gov ). The original Western blot data are provided in Supplementary Materials (Original Western blots).
Kirmani BF, Jacobowitz DM, Namboodiri MAA. Developmental increase of aspartoacylase in oligodendrocytes parallels CNS myelination. Dev Brain Res. 2003;140:105–15.
Article CAS Google Scholar
Moffett JR, Ross B, Arun P, Madhavarao CN, Namboodiri MAA. N-acetylaspartate in the CNS: from neurodiagnostics to neurobiology. Prog Neurobiol. 2007;81:89–131.
Article CAS PubMed PubMed Central Google Scholar
Schuff N, Meyerhoff DJ, Mueller S, Chao L, Sacrey DT, Laxer K, et al. N-acetylaspartate as a marker of neuronal injury in neurodegenerative disease. Adv Exp Med Biol. 2006;576:241–363.
Bogner-Strauss JG. N-acetylaspartate metabolism outside the brain: lipogenesis, histone acetylation, and cancer. Front Endocrinol. 2017;8:240.
Article Google Scholar
Sommer A, Sass JO. Expression of aspartoacylase (ASPA) and Canavan disease. Gene. 2012;505:206–10.
Article CAS PubMed Google Scholar
Mukund K, Subramaniam S. Skeletal muscle: a review of molecular structure and function, in health and disease. Wiley Interdiscip Rev Syst Biol Med. 2020;12:e1462.
Article PubMed Google Scholar
Shefner JM, Musaro A, Ngo ST, Lunetta C, Steyn FJ, Robitaille R, et al. Skeletal muscle in amyotrophic lateral sclerosis. Brain. 2023;146:4425–36.
Article PubMed PubMed Central Google Scholar
Schiaffino S, Reggiani C. Fiber types in mammalian skeletal muscles. Physiol Rev. 2011;91:1447–531.
Peggion C, Massimino ML, Biancotto G, Angeletti R, Reggiani C, Sorgato MC, et al. Absolute quantification of myosin heavy chain isoforms by selected reaction monitoring can underscore skeletal muscle changes in a mouse model of amyotrophic lateral sclerosis. Anal Bioanal Chem. 2017;409:2143–53.
Scaricamazza S, Salvatori I, Giacovazzo G, Loeffler JP, Renè F, Rosina M, et al. Skeletal-muscle metabolic reprogramming in ALS-SOD1G93A mice predates disease onset and is a promising therapeutic target. iScience. 2020;23:101087.
Simone IL, Ruggieri M, Tortelli R, Ceci E, D’Errico E, Leo A, et al. Serum N-acetylaspartate level in amyotrophic lateral sclerosis. Arch Neurol. 2011;68:1308–12.
Spinazzi M, Casarin A, Pertegato V, Salviati L, Angelini C. Assessment of mitochondrial respiratory chain enzymatic activities on tissues and cultured cells. Nat Protoc. 2012;7:1235–46.
Pessentheiner AR, Pelzmann HJ, Walenta E, Schweiger M, Groschner LN, Graier WF, et al. NAT8L (N-Acetyltransferase 8-Like) accelerates lipid turnover and increases energy expenditure in brown adipocytes. J Biol Chem. 2013;288:36040.
Morales PE, Bucarey JL, Espinosa A. Muscle lipid metabolism: role of lipid droplets and perilipins. J Diabetes Res. 2017;2017:1789395.
Qaisar R, Larsson L. What determines myonuclear domain size? Indian J Physiol Pharmacol. 2014;58:1–12.
CAS PubMed Google Scholar
Krause N, Wegner A. N-acetyl-aspartate metabolism at the interface of cancer, immunity, and neurodegeneration. Curr Opin Biotechnol. 2024;85:103051.
Sandri M, Lin J, Handschin C, Yang W, Arany ZP, Lecker SH, et al. PGC-1α protects skeletal muscle from atrophy by suppressing FoxO3 action and atrophy-specific gene transcription. Proc Natl Acad Sci USA 2006;103:16260–5.
Ferri A, Coccurello R. What is “Hyper” in the ALS hypermetabolism? Mediators Inflamm. 2017;2017:7821672.
Jacobs RA, Díaz V, Soldini L, Haider T, Thomassen M, Nordsborg NB, et al. Fast-twitch glycolytic skeletal muscle is predisposed to age-induced impairments in mitochondrial function. J Gerontol A Biol Sci Med Sci. 2013;68:1010–22.
Ciccarone F, Castelli S, Lazzarino G, Scaricamazza S, Mangione R, Bernardini S, et al. Lipid catabolism and mitochondrial uncoupling are stimulated in brown adipose tissue of amyotrophic lateral sclerosis mouse models. Genes Dis. 2023;10:321–4.
Missaglia S, Coleman RA, Mordente A, Tavian D. Neutral lipid storage diseases as cellular model to study lipid droplet function. Cells. 2019;8:187.
Liu L, Fu C, Li F. Acetate affects the process of lipid metabolism in rabbit liver, skeletal muscle and adipose tissue. Animals. 2019;9:799.
Rocchi A, Milioto C, Parodi S, Armirotti A, Borgia D, Pellegrini M, et al. Glycolytic-to-oxidative fiber-type switch and mTOR signaling activation are early-onset features of SBMA muscle modified by high-fat diet. Acta Neuropathol. 2016;132:127–44.
Pereyra AS, Lin CT, Sanchez DM, Laskin J, Spangenburg EE, Neufer PD, et al. Skeletal muscle undergoes fiber type metabolic switch without myosin heavy chain switch in response to defective fatty acid oxidation. Mol Metab. 2022;59:101456.
Zhang L, Zhou Y, Wu W, Hou L, Chen H, Zuo B, et al. Skeletal muscle-specific overexpression of PGC-1α induces fiber-type conversion through enhanced mitochondrial respiration and fatty acid oxidation in mice and pigs. Int J Biol Sci. 2017;13:1152–62.
Lin J, Wu H, Tarr PT, Zhang CY, Wu Z, Boss O, et al. Transcriptional co-activator PGC-1 alpha drives the formation of slow-twitch muscle fibres. Nature. 2002;418:797–801.
Baldelli S, Aquilano K, Ciriolo MR. PGC-1α buffers ROS-mediated removal of mitochondria during myogenesis. Cell Death Dis. 2014;5:e1515.
Johri A, Chandra A, Beal MF. PGC-1α, mitochondrial dysfunction and Huntington’s disease. Free Radic Biol Med. 2013;62:37–46.
Stein TP, Wade CE. Metabolic consequences of muscle disuse atrophy. J Nutr. 2005;135:1824S–28S.
Davies MR, Garcia S, Liu M, Chi H, Kim HT, Raffai RL, et al. Muscle-derived beige adipose precursors secrete promyogenic exosomes that treat rotator cuff muscle degeneration in mice and are identified in humans by single-cell RNA sequencing. Am J Sports Med. 2022;50:2247–57.
Download references
Acknowledgements
LL, FF and ADC are enrolled in PhD Program in Cellular and Molecular Biology, Department of Biology, University of Rome Tor Vergata, Rome, Italy.
This work was partially supported by: Beyond Borders (E84I20000580005 to MRC) and Ricerca Scientifica d’Ateneo 2021 (E83C22000160005 to FC) University of Rome Tor Vergata; Italian Ministry of Health - Ricerca Corrente; Italian Ministry of Health - Ricerca Finalizzata Giovani Ricercatori GR-2019-12369983; #NEXTGENERATIONEU (NGEU) funded by the Ministry of University and Research (MUR), National Recovery and Resilience Plan (NRRP), project MNESYS (PE0000006) – (DN. 1553 11.10.2022).
Author information
These authors contributed equally: Maria Rosa Ciriolo, Fabio Ciccarone.
Authors and Affiliations
IRCCS San Raffaele Roma, Rome, Italy
Serena Castelli, Maria Rosa Ciriolo & Fabio Ciccarone
Department of Human Sciences and Promotion of the Quality of Life, San Raffaele Open University, Rome, Italy
Enrico Desideri
Department of Biology, University of Rome Tor Vergata, Rome, Italy
Leonardo Laureti, Federica Felice, Angela De Cristofaro, Maria Rosa Ciriolo & Fabio Ciccarone
IRCCS Fondazione Santa Lucia, Rome, Italy
Silvia Scaricamazza
UniCamillus-Saint Camillus International University of Health and Medical Sciences, Rome, Italy
Giacomo Lazzarino
You can also search for this author in PubMed Google Scholar
Contributions
SC designed and managed the experiments, performed the statistical analysis, conceptualized data and wrote the original draft; LL performed and analyzed some of the Western blot experiments; FF and ADC provided methodological support; ED performed the bioinformatic analysis; SS was involved in SOD1-G93A mouse colony management including breeding, genotyping and evaluation of symptoms during disease progression; GL performed HPLC analysis; FC and MRC performed study concept and design, edited and reviewed the manuscript and acquired funding. FC and MRC equally contributed as co-last authors. All authors read and approved the final manuscript.
Corresponding author
Correspondence to Fabio Ciccarone .
Ethics declarations
Competing interests.
The authors declare no competing interests.
Ethics approval
All animal care and experimental procedures were conducted in accordance with Animal Welfare Office, Department of Public Health and Veterinary, Nutrition and Food Safety, General Management of Animal Care and Veterinary Drugs of the Italian Ministry of Health (protocol number 931/2017/PR) and performed following the European Guidelines for the use of animals in research (2010/63/EU) and the requirements of Italian laws (D.L. 26/2014).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Mauro Piacentini
Supplementary information
Supplemental materials, original western blot images, rights and permissions.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ .
Reprints and permissions
About this article
Cite this article.
Castelli, S., Desideri, E., Laureti, L. et al. N-acetylaspartate promotes glycolytic-to-oxidative fiber-type switch and resistance to atrophic stimuli in myotubes. Cell Death Dis 15 , 686 (2024). https://doi.org/10.1038/s41419-024-07047-0
Download citation
Received : 09 April 2024
Revised : 23 August 2024
Accepted : 02 September 2024
Published : 19 September 2024
DOI : https://doi.org/10.1038/s41419-024-07047-0
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies

IMAGES
VIDEO
COMMENTS
Simply put, lateral excursion is when the mandible moves to one side. But the mechanism behind this is not as simple as it sounds. The mandible is capable of two movements if we take the condyle as the hinge: rotation and translation. Imagine a vertical and horizontal axis passing through the condyles. The movement of the mandible with respect ...
Excursion can occur in either direction, and anatomists use directional terms to specify the type of excursion. When the mandible moves to either the left or right, it's moving away from the body's midline, so it's called lateral excursion. When the mandible moves closer to the midline of the body, it's called medial excursion.
The degree and type of movement that can be produced at a synovial joint is determined by its structural type. While the ball-and-socket joint gives the greatest range of movement at an individual joint, in other regions of the body, several joints may work together to produce a particular movement. ... Lateral excursion moves the mandible away ...
The type of movement that can be produced at a synovial joint is determined by its structural type. While the ball-and-socket joint gives the greatest range of movement at an individual joint, in other regions of the body, several joints may work together to produce a particular movement. ... Lateral excursion moves the mandible away from the ...
The type of movement that can be produced at a synovial joint is determined by its structural type. While the ball-and-socket joint gives the greatest range of movement at an individual joint, in other regions of the body, several joints may work together to produce a particular movement. ... lateral excursion side-to-side movement of the ...
The type of movement that can be produced at a synovial joint is determined by its structural type. While the ball-and-socket joint gives the greatest range of movement at an individual joint, in other regions of the body, several joints may work together to produce a particular movement. ... Lateral excursion moves the mandible away from the ...
The type of movement that can be produced at a synovial joint is determined by its structural type. While the ball-and-socket joint gives the greatest range of movement at an individual joint, in other regions of the body, several joints may work together to produce a particular movement. ... Lateral excursion moves the mandible away from the ...
Lateral excursion. Lateral excursions are a form of dynamic occlusion which occurs when the mandible moves left or right with teeth in contact. They can be described as: Canine guidance: canine protected articulation. The canines on the working side are the only occluding teeth whilst all other teeth become discluded when carrying out lateral ...
Types of anterior guidance in lateral excursions. Click the pictures to see typical ink marks associated with each type of guidance. Note the dots formed by intercuspal position (static), and the lines caused by a lateral excursion to the right.
In these lateral excursions, only the canines contact and take the occlusal load, while the other teeth are separated. The canines are better suited than the posterior teeth to distribute these sideways forces for several reasons. Their shape is strong enough to resist force unlike molar teeth that have fissures causing weaknesses.
Lateral: 8 to 12 mm. Protrusive: 8 to 12 mm. Opening Movements. Place the notch of the range of motion scale on the midline of the mandibular centrals, and take the following three measurements: Comfort: ask the patient to open "comfortably without pain.". Active: ask the patient to open as wide as they can even if it hurts.
Normal ROM is: 45 mm for depression, lateral excursion is 1/4 of depression, protrusion is 6-9 mm and retrusion is 3 mm (Ho, 2011). Lateral deviation to one side may signify capsular restrictions ipsilaterally, potential muscle dysfunction, or an anteriorly displaced disc without reduction ipsilaterally. This may be represented as a "C-curve ...
Excursion can occur in either direction, and anatomists use directional terms to specify the type of excursion. When the mandible moves to either the left or right, it's moving away from the body's midline, so it's called lateral excursion. When the mandible moves closer to the midline of the body, it's called medial excursion.
The functional motions permitted are mandibular depression (mouth opening), mandibular elevation (mouth closing), protrusion (anterior translation; Fig. 13.3) and retrusion (posterior translation; Fig. 13.4), and right and left lateral excursion or laterotrusion (lateral deviation; Fig. 13.5). Maximal contact of the teeth in mouth closing is ...
TMJ Movements. Normal movements of the jaw during function, such as chewing, are known as excursions. There are two lateral excursions ( left and right ) and the forward excursion, known as protrusion, the reversal of which is retrusion. When the jaw is moved into protrusion, the lower incisors or front teeth are moved so that they first come ...
The lateral excursion is the movement to the left and right of the zero position of the jaw. The medial excursion is the movement back to the zero position of the jaw (Saladin, pg. 291). We can experience jaw pain from many different sources. Some of the more traumatic forms of a jaw injury can occur with a deep yawn or a strenuous depression ...
The type of movement that can be produced at a synovial joint is determined by its structural type. While the ball-and-socket joint gives the greatest range of movement at an individual joint, in other regions of the body, several joints may work together to produce a particular movement. ... Lateral excursion moves the mandible away from the ...
During a lateral excursion to the right, the condyle and the disk on the left side glide inferiorly, anteriorly, and laterally in the sagittal plane and medially in the horizontal plane along the articular eminence. ... and sinusitis. 70 Patients with sinusitis complain of acute facial pain or pressure type headaches, and may present with nasal ...
rotation) of the mandibular condyle, while lateral excursion occurs with ipsilateral rotation and contralateral translation of the mandibular condyles.8 Restoration of these normal accessory motions and joint mechanics is important in the rehabilitation of the TMJ in order to restore functional movements of the jaw.
The type of guidance at the 0.5 mm position was not significantly associated with the incisor (Table 1), canine (Table 2), or molar classification (Table 3). However, it should be noted that at 0.5 mm lateral excursion, bilateral canine protected occlusion was observed only in subjects with Class I incisor, canine, or molar relationships
Notably, glycolytic-to-oxidative fiber-type switch has been observed in motor neuron diseases such as amyotrophic lateral sclerosis (ALS), where muscles undergo an increase in lipid oxidative ...